NTA JEE Main 2nd September 2020 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 26
The number of subshells associated with $$n = 4$$ and $$m = -2$$ quantum numbers is:
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 27
Three elements X, Y and Z are in the 3$$^{rd}$$ period of the periodic table. The oxides of X, Y and Z, respectively, are basic, amphoteric and acidic. The correct order of the atomic numbers of X, Y and Z is:
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 28
The shape/structure of $$[XeF_5]^-$$ and $$XeO_3F_2$$, respectively are:
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 29
Match the type of interaction in column A with the distance dependence of their interaction energy in column B:
A B
(i) ion - ion (a) $$\frac{1}{r}$$
(ii) Dipole - dipole (b) $$\frac{1}{r^2}$$
(iii) London dispersion (c) $$\frac{1}{r^3}$$
(iv) $$\frac{1}{r^6}$$
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 30
The molecular geometry of $$SF_6$$ is octahedral. What is the geometry of $$SF_4$$ (including lone pair(s) of electrons, if any)?
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 31
Two elements A and B have similar chemical properties. They don't form solid hydrogencarbonates, but react with nitrogen to form nitrides. A and B, respectively, are:
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 32
Arrange the following labelled hydrogens in decreasing order of acidity: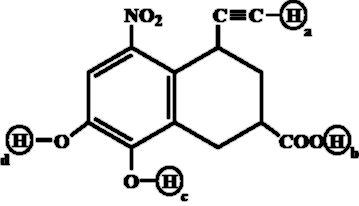
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 33
An organic compound 'A' $$(C_9H_{10}O)$$ when treated with conc. HI undergoes cleavage to yield compound 'B' and 'C'. 'B' gives yellow precipitate with $$AgNO_3$$ where as 'C' tautomerizes to 'D'. 'D' gives positive iodoform test. 'A' could be:
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 34
The major product of the following reaction is:
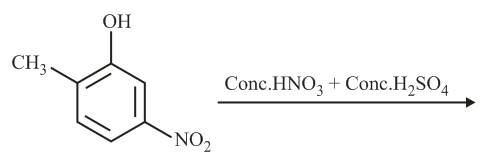
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 35
The size of a raw mango shrinks to a much smaller size when kept in a concentrated salt solution. Which one of the following process can explain this?
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 36
The results given in the below table were obtained during kinetic studies of the following reaction:
$$2A + B \to C + D$$
| Experiment | [A]/mol L$$^{-1}$$ | [B]/mol L$$^{-1}$$ | Initial rate/mol L$$^{-1}$$ min$$^{-1}$$ |
|---|---|---|---|
| I | 0.1 | 0.1 | $$6.00 \times 10^{-3}$$ |
| II | 0.1 | 0.2 | $$2.40 \times 10^{-2}$$ |
| III | 0.2 | 0.1 | $$1.20 \times 10^{-2}$$ |
| IV | X | 0.2 | $$7.20 \times 10^{-2}$$ |
| V | 0.3 | Y | $$2.88 \times 10^{-1}$$ |
X and Y in the given table are respectively:
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 37
Amongst the following statements regarding adsorption, those that are valid are:
(a) $$\Delta H$$ becomes less negative as adsorption proceeds.
(b) On a given adsorbent, ammonia is adsorbed more than nitrogen gas.
(c) On adsorption, the residual force acting along the surface of the adsorbent increases
(d) With increase in temperature, the equilibrium concentration of adsorbate increases.
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 38
Cast iron is used for the manufacture of:
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 39
Simplified absorption spectra of three complexes ((i) and (ii) and (iii)) of $$M^{+n}$$ ion are provided below; their $$\lambda_{max}$$ values are marked as A, B and C respectively. The correct match between the complexes and their $$\lambda_{max}$$ values is: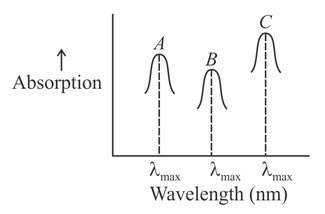
(i) $$[M(NCS)_6]^{(-6+n)}$$
(ii) $$[MF_6]^{(-6+n)}$$
(iii) $$[M(NH_3)_6]^{n+}$$
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 40
The one that is not expected to show isomerism is:
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 41
The major product obtained from $$E_2$$-elimination of 3-bromo-2-fluoropentane is:
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 42
Consider the reaction sequence given below:
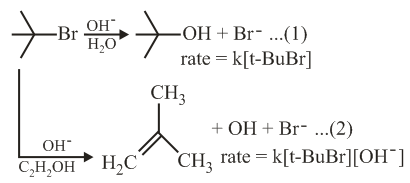
Which of the following statements is true:
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 43
Two compounds A and B with same molecular formula $$(C_3H_6O)$$ undergo Grignard reaction with methylmagnesium bromide to give products C and D. Products C and D show following chemical tests:
| Test | C | D |
|---|---|---|
| Ceric ammonium nitrate Test | Positive | Positive |
| Lucas Test | Turbidity obtained after five minutes | Turbidity obtained Immediately |
| Iodoform Test | Positive | Negative |
C and D respectively are:
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 44
If you spill a chemical toilet cleaning liquid on your hand, your first aid would be:
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 45
The correct observation in the following reaction is:
$$Sucrose \xrightarrow[Cleavage \; (Hydrolysis)]{Glycosidic \; bond \; } A + B \xrightarrow[ Reagent]{Seliwanoff } ?$$
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 46
The ratio of the mass percentages of 'C & H' and 'C & O' of a saturated acyclic organic compound 'X' are 4 : 1 and 3 : 4 respectively. Then, the moles of oxygen gas required for complete combustion of two moles of organic compound 'X' is ___________.
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 47
The work function of sodium metal is $$4.41 \times 10^{-19}$$ J. If photons of wavelength 300 nm are incident on the metal, the kinetics energy of the ejected electrons will be $$(h = 6.63 \times 10^{-34}$$ J s; $$c = 3 \times 10^8$$ m s$$^{-1})$$ ___________ $$\times 10^{-21}$$ J.
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 48
The heat of combustion of ethanol into carbon dioxides and water is $$-327$$ Kcal at constant pressure. The heat evolved (in cal) at constant volume at 27$$°$$C (if all gases behave ideally) is $$(R = 2$$ cal mol$$^{-1}$$ K$$^{-1})$$ ___________.
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 49
The oxidation states of transition metal atoms in $$K_2Cr_2O_7$$, $$KMnO_4$$ and $$K_2FeO_4$$, respectively, are x, y and z. The sum of x, y and z is ___________.
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 2 - Chemistry - Question 50
For the disproportionation reaction $$2Cu^+(aq) \rightleftharpoons Cu(s) + Cu^{2+}(aq)$$ at 298K, ln K (where K is the equilibrium constant) is ___________ $$\times 10^{-1}$$.
Given: $$(E^°_{Cu^{2+}/Cu^+} = 0.16V, E^°_{Cu^+/Cu} = 0.52V, \frac{RT}{F} = 0.025)$$
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




