NTA JEE Main 2nd September 2020 Shift 1
For the following questions answer them individually
NTA JEE Main 2nd September 2020 Shift 1 - Question 41
Consider that $$d^6$$ metal ion $$(M^{2+})$$ forms a complex with aqua ligands, and the spin only magnetic moment of the complex is 4.90 BM. The geometry and the crystal field stabilization energy of the complex is:
NTA JEE Main 2nd September 2020 Shift 1 - Question 42
For octahedral Mn(II) and tetrahedral Ni(II) complexes, consider the following statements:
(I) both the complexes can be high spin.
(II) Ni(II) complex can very rarely be of low spin.
(III) with strong field ligands, Mn(II) complexes can be low spin.
(IV) aqueous solution of Mn(II) ions is yellow in color.
The correct statements are:
NTA JEE Main 2nd September 2020 Shift 1 - Question 43
Which of the following compounds will show retention in configuration on nucleophilic substitution by $$OH^-$$ ion?
NTA JEE Main 2nd September 2020 Shift 1 - Question 44
The major aromatic product C in the following reaction sequence will be:
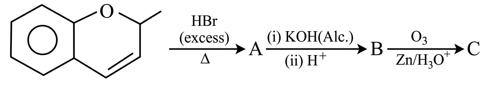
NTA JEE Main 2nd September 2020 Shift 1 - Question 45
Consider the following reactions:
(i) Glucose + ROH $$\xrightarrow{dry \; HCl}$$ Acetal $$\xrightarrow{x \; eq. \; of \; (CH_3CO)_2O}$$ acetyl derivative
(ii) Glucose $$\xrightarrow{Ni/H_2}$$ A $$\xrightarrow{y \; eq. \; of \; (CH_3CO)_2O}$$ acetyl derivative
(iii) Glucose $$\xrightarrow{z \; eq. \; of \; (CH_3CO)_2O}$$ acetyl derivative
'x', 'y' and 'z' in these reactions are respectively:
NTA JEE Main 2nd September 2020 Shift 1 - Question 46
The Gibbs energy change (in J) for the given reaction at $$[Cu^{2+}] = [Sn^{2+}] = 1$$ M and 298 K is:
$$Cu(s) + Sn^{2+}(aq.) \to Cu^{2+}(aq.) + Sn(s)$$;
$$(E^0_{Sn^{2+}|Sn} = -0.16 \; V, E^0_{Cu^{2+}|Cu} = 0.34 \; V, \text{Take } F = 96500 \; C \; mol^{-1})$$
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 1 - Question 47
The internal energy change (in J) when 90 g of water undergoes complete evaporation at 100$$°$$C is ___________.
(Given: $$\Delta H_{vap}$$ for water at 373 K $$= 41$$ kJ/mol, R $$= 8.314$$ J K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 1 - Question 48
The number of chiral carbons present in the molecule given below is ___________.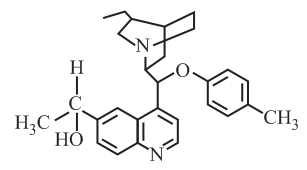
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 1 - Question 49
The mass of gas adsorbed, x, per unit mass of adsorbate, m, was measured at various pressures, p. A graph between $$\log \frac{x}{m}$$ and $$\log p$$ gives a straight line with slope equal to 2 and the intercept equal to 0.4771. The value of $$\frac{x}{m}$$ at a pressure of 4 atm is:
(Given $$\log 3 = 0.4771$$)
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 1 - Question 50
The oxidation states of iron atoms in compounds (A), (B) and (C), respectively, are x, y and z. Then sum of x, y and z is ___________.
(A) $$Na_4[Fe(CN)_5(NOS)]$$ (B) $$Na_4[FeO_4]$$ (C) $$[Fe_2(CO)_9]$$
789
456
123
0.-
Clear All


