NTA JEE Main 18th March 2021 Shift 2
For the following questions answer them individually
NTA JEE Main 18th March 2021 Shift 2 - Question 51
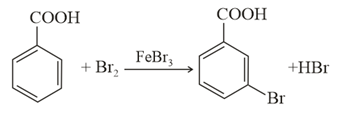
Consider the above reaction where 6.1 g of benzoic acid is used to get 7.8 g of m-bromo benzoic acid. The percentage yield of the product is ___.
(Round off to the Nearest integer)
[Given : Atomic masses : C = 12.0 u, H : 1.0 u, O : 16.0 u, Br = 80.0 u]
789
456
123
0.-
Clear All
NTA JEE Main 18th March 2021 Shift 2 - Question 52
The number of species below that have two lone pairs of electrons in their central atom is ___ (Round off to the Nearest integer)
SF$$_4$$, BF$$_4^-$$, ClF$$_3$$, AsF$$_3$$, PCl$$_5$$, BrF$$_5$$, XeF$$_4$$, SF$$_6$$
789
456
123
0.-
Clear All
NTA JEE Main 18th March 2021 Shift 2 - Question 53
The gas phase reaction
$$2 A(g) \rightleftharpoons A_2(g)$$
at 400 K has $$\Delta G^\circ = +25.2$$ kJ mol$$^{-1}$$.
The equilibrium constant $$K_C$$ for this reaction is ___ $$\times 10^{-2}$$. (Round off to the Nearest integer)
Use : $$R = 8.3$$ J mol$$^{-1}$$ K$$^{-1}$$, ln 10 = 2.3, $$\log_{10} 2 = 0.30$$, 1 atm = 1 bar
antilog(-0.3) = 0.501
789
456
123
0.-
Clear All
NTA JEE Main 18th March 2021 Shift 2 - Question 54
The solubility of CdSO$$_4$$ in water is $$8.0 \times 10^{-4}$$ mol L$$^{-1}$$. Its solubility in 0.01 M H$$_2$$SO$$_4$$ solution is ___ $$\times 10^{-6}$$ mol L$$^{-1}$$ (Round off to the Nearest integer) (Assume that solubility is much less than 0.01 M)
789
456
123
0.-
Clear All
NTA JEE Main 18th March 2021 Shift 2 - Question 55
10.0 ml of Na$$_2$$CO$$_3$$ solution is titrated against 0.2 M HCl solution. The following values were obtained in 5 readings. 4.8 ml, 4.9 ml, 5.0 ml, 5.0 ml and 5.0 ml
Based on these readings, and convention of titrimetric estimation of concentration of Na$$_2$$CO$$_3$$ solution is ___ mM.
(Round off to the Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 18th March 2021 Shift 2 - Question 56
A solute A dimerizes in water. The boiling point of a 2 molal solution of A is 100.52$$^\circ$$C. The percentage association of A is ___.
(Round off to the Nearest integer)
Use : K$$_b$$ for water = 0.52 K kg mol$$^{-1}$$
Boiling point of water = 100$$^\circ$$C
789
456
123
0.-
Clear All
NTA JEE Main 18th March 2021 Shift 2 - Question 57
The molar conductivity at infinite dilution of barium chloride, sulphuric acid and hydrochloric acid are 280, 860, 426 Scm$$^2$$mol$$^{-1}$$ respectively. The molar conductivity at infinite dilution of barium sulphate is ___ Scm$$^2$$ mol$$^{-1}$$ (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 18th March 2021 Shift 2 - Question 58
A reaction has a half life of 1 min. The time required for 99.9% completion of the reaction is ___ min.
(Round off to the Nearest integer)
[Use : ln 2 = 0.69, ln 10 = 2.3]
789
456
123
0.-
Clear All
NTA JEE Main 18th March 2021 Shift 2 - Question 59
A xenon compound A upon partial hydrolysis gives XeO$$_2$$F$$_2$$. The number of lone pair of electrons present in compound A is ___ (Round off to the Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 18th March 2021 Shift 2 - Question 60
In Tollen's test for aldehyde, the overall number of electron(s) transferred to the Tollen's reagent formula [Ag(NH$$_3$$)$$_2$$]$$^+$$ per aldehyde group to form silver mirror is ___. (Round off to the Nearest integer)
789
456
123
0.-
Clear All


