NTA JEE Main 17th March 2021 Shift 2
For the following questions answer them individually
NTA JEE Main 17th March 2021 Shift 2 - Question 51
Consider the above reaction. The percentage yield of amide product is ________ (Round off to the Nearest Integer).
(Given: Atomic mass: C: 12.0u, H: 1.0u, N: 14.0u, O: 16.0u, Cl: 35.5u)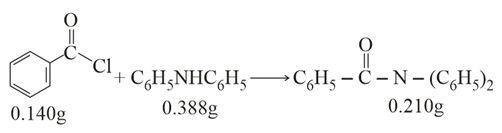
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Question 52
The number of chlorine atoms in 20 mL of chlorine gas at STP is ________ $$\times 10^{21}$$. (Round off to the Nearest Integer).
[Assume chlorine is an ideal gas at STP. R = 0.083 L bar mol$$^{-1}$$ K$$^{-1}$$, $$N_A = 6.023 \times 10^{23}$$]
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Question 53
Consider the reaction $$N_2O_4(g) \rightleftharpoons 2NO_2(g)$$. The temperature at which $$K_C = 20.4$$ and $$K_P = 600.1$$, is ________ K. (Round off to the Nearest Integer). [Assume all gases are ideal and R = 0.0831 L bar K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Question 54
The total number of C-C sigma bond/s in mesityl oxide ($$C_6H_{10}O$$) is ________ (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Question 55
KBr is doped with $$10^{-5}$$ mole percent of SrBr$$_2$$. The number of cationic vacancies in 1 g of KBr crystal is $$10^{14}$$ ________. (Round off to the Nearest Integer). [Atomic Mass: K: 39.1u, Br: 79.9u, $$N_A = 6.023 \times 10^{23}$$]
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Question 56
A 1 molal $$K_4Fe(CN)_6$$ solution has a degree of dissociation of 0.4. Its boiling point is equal to that of another solution which contains 18.1 weight percent of a non electrolytic solute A. The molar mass of A is ________ u. (Round off to the Nearest Integer). [Density of water = 1.0 g cm$$^{-3}$$]
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Question 57
A KCl solution of conductivity 0.14 S m$$^{-1}$$ shows a resistance of 4.19$$\Omega$$ in a conductivity cell. If the same cell is filled with an HCl solution, the resistance drops to 1.03$$\Omega$$. The conductivity of the HCl solution is ________ $$\times 10^{-2}$$ S m$$^{-1}$$. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Question 58
The reaction $$2A + B_2 \to 2AB$$ is an elementary reaction. For a certain quantity of reactants, if the volume of the reaction vessel is reduced by a factor of 3, the rate of the reaction increases by a factor of ________. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Question 59
In the ground state of atomic Fe (Z = 26), the spin-only magnetic moment is ________ $$\times 10^{-1}$$ BM. (Round off to the Nearest Integer).
[Given: $$\sqrt{3} = 1.73, \sqrt{2} = 1.41$$]
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Question 60
On complete reaction of FeCl$$_3$$ with oxalic acid in aqueous solution containing KOH, resulted in the formation of product A. The secondary valency of Fe in the product A is ________. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All


