NTA JEE Main 17th March 2021 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 31
Amongst the following, the linear species is:
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 32
During which of the following processes, does entropy decrease?
(A) Freezing of water to ice at 0°C
(B) Freezing of water to ice at -10°C
(C) N$$_2$$(g) + 3H$$_2$$(g) $$\to$$ 2NH$$_3$$(g)
(D) Adsorption of CO(g) and lead surface
(E) Dissolution of NaCl in water
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 33
The functional groups that are responsible for the ion-exchange property of cation and anion exchange resins, respectively, are:
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 34
The set of elements that differ in mutual relationship from those of the other sets is:
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 35
One of the by-products formed during the recovery of NH$$_3$$ from Solvay process is:
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 36
The correct pair(s) of the ambident nucleophiles is (are):
(A) AgCN / KCN
(B) RCOOAg / RCOOK
(C) AgNO$$_2$$ / KNO$$_2$$
(D) AgI / KI
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 37
Nitrogen can be estimated by Kjeldahl's method for which of the following compound?
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 38
Given below are two statements:
Statement-I: 2-methylbutane on oxidation with KMnO$$_4$$ gives 2-methylbutan-2-ol.
Statement-II: n-alkanes can be easily oxidised to corresponding alcohol with KMnO$$_4$$.
Choose the correct option:
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 39
Choose the correct statement regarding the formation of carbocations A and B given:
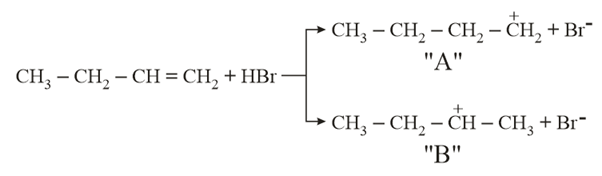
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 40
Which of the following statement(s) is (are) incorrect reason for eutrophication?
(A) excess usage of fertilisers
(B) excess usage of detergents
(C) dense plant population in water bodies
(D) lack of nutrients in water bodies that prevent plant growth
Choose the most appropriate answer from the options given below:
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 41
For the coagulation of a negative sol, the species below, that has the highest flocculating power is:
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 42
Match List-I and List-II:
List-I List-II
a. Haematite i. Al$$_2$$O$$_3$$ . xH$$_2$$O
b. Bauxite ii. Fe$$_2$$O$$_3$$
c. Magnetite iii. CuCO$$_3$$ . Cu(OH)$$_2$$
d. Malachite iv. Fe$$_3$$O$$_4$$
Choose the correct answer from the options given below:
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 43
The set that represents the pair of neutral oxides of nitrogen is:
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 44
The common positive oxidation states for an element with atomic number 24, are:
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 45
Match List-I with List-II:
List-I List-II
a. [Co(NH$$_3$$)$$_6$$][Cr(CN)$$_6$$] i. Linkage isomerism
b. [Co(NH$$_3$$)$$_3$$(NO$$_2$$)$$_3$$] ii. Solvate isomerism
c. [Cr(H$$_2$$O)$$_6$$]Cl$$_3$$ iii. Co-ordination isomerism
d. $$cis$$-[CrCl$$_2$$(ox)$$_2$$]$$^{3-}$$ iv. Optical isomerism
Choose the correct answer from the options given below:
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 46
In the above reaction, the structural formula of (A), "X" and "Y" respectively are:
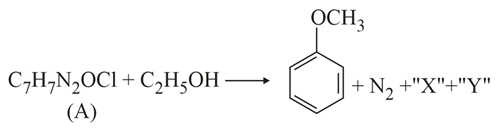
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 47
Primary, secondary and tertiary amines can be separated using:
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 48
Match List-I with List-II:
List-I (Chemical Compound) List-II (Used as)
a. Sucralose i. Synthetic detergent
b. Glyceryl ester of stearic acid ii. Artificial sweetener
c. Sodium benzoate iii. Antiseptic
d. Bithional iv. Food preservative
Choose the correct match:
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 49
Fructose is an example of:
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 50
In the above reactions, the enzyme A and enzyme B respectively are:
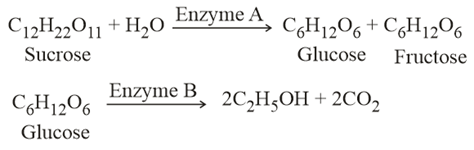
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 51
Consider the above reaction. The percentage yield of amide product is ________ (Round off to the Nearest Integer).
(Given: Atomic mass: C: 12.0u, H: 1.0u, N: 14.0u, O: 16.0u, Cl: 35.5u)
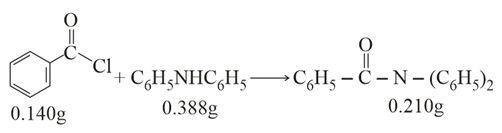
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 52
The number of chlorine atoms in 20 mL of chlorine gas at STP is ________ $$\times 10^{21}$$. (Round off to the Nearest Integer).
[Assume chlorine is an ideal gas at STP. R = 0.083 L bar mol$$^{-1}$$ K$$^{-1}$$, $$N_A = 6.023 \times 10^{23}$$]
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 53
Consider the reaction $$N_2O_4(g) \rightleftharpoons 2NO_2(g)$$. The temperature at which $$K_C = 20.4$$ and $$K_P = 600.1$$, is ________ K. (Round off to the Nearest Integer). [Assume all gases are ideal and R = 0.0831 L bar K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 54
The total number of C-C sigma bond/s in mesityl oxide ($$C_6H_{10}O$$) is ________ (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 55
KBr is doped with $$10^{-5}$$ mole percent of SrBr$$_2$$. The number of cationic vacancies in 1 g of KBr crystal is $$10^{14}$$ ________. (Round off to the Nearest Integer). [Atomic Mass: K: 39.1u, Br: 79.9u, $$N_A = 6.023 \times 10^{23}$$]
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 56
A 1 molal $$K_4Fe(CN)_6$$ solution has a degree of dissociation of 0.4. Its boiling point is equal to that of another solution which contains 18.1 weight percent of a non electrolytic solute A. The molar mass of A is ________ u. (Round off to the Nearest Integer). [Density of water = 1.0 g cm$$^{-3}$$]
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 57
A KCl solution of conductivity 0.14 S m$$^{-1}$$ shows a resistance of 4.19$$\Omega$$ in a conductivity cell. If the same cell is filled with an HCl solution, the resistance drops to 1.03$$\Omega$$. The conductivity of the HCl solution is ________ $$\times 10^{-2}$$ S m$$^{-1}$$. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 58
The reaction $$2A + B_2 \to 2AB$$ is an elementary reaction. For a certain quantity of reactants, if the volume of the reaction vessel is reduced by a factor of 3, the rate of the reaction increases by a factor of ________. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 59
In the ground state of atomic Fe (Z = 26), the spin-only magnetic moment is ________ $$\times 10^{-1}$$ BM. (Round off to the Nearest Integer).
[Given: $$\sqrt{3} = 1.73, \sqrt{2} = 1.41$$]
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 2 - Chemistry - Question 60
On complete reaction of FeCl$$_3$$ with oxalic acid in aqueous solution containing KOH, resulted in the formation of product A. The secondary valency of Fe in the product A is ________. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




