NTA JEE Main 16th March 2021 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 31
Identify the elements X and Y using the ionization energy values given below:
Ionization energy (kJ/mol):
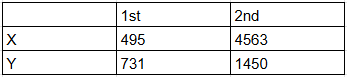
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 32
The characteristics of elements X, Y and Z with atomic numbers, respectively, 33, 53 and 83 are:
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 33
Statement I: Sodium hydride can be used as an oxidising agent.
Statement II: The lone pair of electrons on nitrogen in pyridine makes it basic.
Choose the CORRECT answer from the options given below:
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 34
The exact volumes of 1 M NaOH solution required to neutralise 50 mL of 1 M $$H_3PO_3$$ solution and 100 mL of 2 M $$H_3PO_2$$ solution, respectively, are:
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 35
The correct statements about $$H_2O_2$$ are:
(A) used in the treatment of effluents.
(B) used as both oxidising and reducing agents.
(C) the two hydroxyl groups lie in the same plane.
(D) miscible with water.
Choose the correct answer from the options given below:
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 36
The INCORRECT statement regarding the structure of $$C_{60}$$ is:
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 37
An unsaturated hydrocarbon X on ozonolysis gives A. Compound A when warmed with ammoniacal silver nitrate forms a bright silver mirror along the sides of the test tube. The unsaturated hydrocarbon X is:
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 38
The green house gas/es is (are):
(A) Carbon dioxide
(B) Oxygen
(C) Water vapour
(D) Methane
Choose the most appropriate answer from the options given below:
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 39
The INCORRECT statements below regarding colloidal solutions is:
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 40
Which of the following reduction reaction CANNOT be carried out with coke?
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 41
$$FeX_2$$ and $$FeY_3$$ are known when $$x$$ and $$y$$ are:
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 42
Arrange the following metal complex/compounds in the increasing order of spin only magnetic moment. Presume all the three, high spin system.
(Atomic numbers Ce = 58, Gd = 64 and Eu = 63.)
(a) $$(NH_4)_2[Ce(NO_3)_6]$$
(b) $$Gd(NO_3)_3$$
(c) $$Eu(NO_3)_3$$
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 43
Identify the reagent(s) 'A' and condition(s) for the reaction:
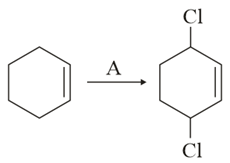
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 44
In the above reaction, the reagent "A" is:
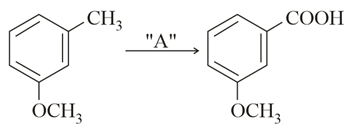
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 45
Ammonolysis of Alkyl halides followed by the treatment with NaOH solution can be used to prepare primary, secondary and tertiary amines. The purpose of NaOH in the reaction is.
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 46
Which of the following is least basic?
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 47
The structure of X is:
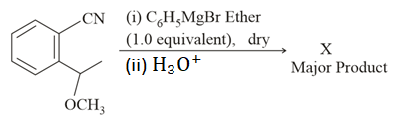
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 48
Which of the following polymer is used in the manufacture of wood laminates?
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 49
Match List-I with List-II:

The correct match is:
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 50
The secondary structure of protein is stabilised by:
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 51
When 35 mL of 0.15 M lead nitrate solution is mixed with 20 mL of 0.12 M chromic sulphate solution, ________ $$\times 10^{-5}$$ moles of lead sulphate precipitate out. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 52
The number of orbitals with $$n = 5$$, $$m_l = +2$$ is ________. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 53
At 25°C, 50 g of iron reacts with HCl to form $$FeCl_2$$. The evolved hydrogen gas expands against a constant pressure of 1 bar. The work done by the gas during this expansion is ________ J. (Round off to the Nearest Integer)
[Given: $$R = 8.314$$ J mol$$^{-1}$$ K$$^{-1}$$. Assume, hydrogen is an ideal gas]
[Atomic mass of Fe is 55.85 u]
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 54
Sulphurous acid ($$H_2SO_3$$) has $$Ka_1 = 1.7 \times 10^{-2}$$ and $$Ka_2 = 6.4 \times 10^{-8}$$. The pH of 0.588 M $$H_2SO_3$$ is ________. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 55
In Duma's method of estimation of nitrogen, 0.1840 g of an organic compound gave 30 mL of nitrogen collected at 287 K and 758 mm of Hg pressure. The percentage composition of nitrogen in the compound is ________. (Round off to the Nearest Integer).
[Given: Aqueous tension at 287 K = 14 mm of Hg]
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 56
Ga (atomic mass 70 u) crystallizes in a hexagonal close packed structure. The total number of voids in 0.581 g of Ga is ________ $$\times 10^{21}$$. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 57
At 363 K, the vapour pressure of A is 21 kPa and that of B is 18 kPa. One mole of A and 2 moles of B are mixed. Assuming that this solution is ideal, the vapour pressure of the mixture is ________ kPa. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 58
A 5.0 m mol dm$$^{-3}$$ aqueous solution of KCl has a conductance of 0.55 mS when measured in a cell constant 1.3 cm$$^{-1}$$. The molar conductivity of this solution is ________ mS m$$^2$$ mol$$^{-1}$$. (Round off to the Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 59
A and B decompose via first order kinetics with half-lives 54.0 min and 18.0 min respectively. Starting from an equimolar non-reactive mixture of A and B, the time taken for the concentration of A to become 16 times that of B is ________ min. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 2 - Chemistry - Question 60
$$[Ti(H_2O)_6]^{3+}$$ absorbs light of wavelength 498 nm during a d-d transition. The octahedral splitting energy for the above complex is ________ $$\times 10^{-19}$$ J. (Round off to the Nearest Integer).
$$h = 6.626 \times 10^{-34}$$ Js; $$c = 3 \times 10^8$$ ms$$^{-1}$$.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




