NTA JEE Main 16th March 2021 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 31
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: The H-O-H bond angle in water molecule is 104.5°.
Reason R: The lone pair - lone pair repulsion of electrons is higher than the bond pair - bond pair repulsion.
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 32
Given below are two statements:
Statement I: $$H_2O_2$$ can act as both oxidising and reducing agent in basic medium.
Statement II: In the hydrogen economy, the energy is transmitted in the form of dihydrogen. In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 33
A group 15 element, which is a metal and forms a hydride with strongest reducing power among group 15 hydrides. The element is:
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 34
Given below are two statements:
Statement I: Both $$CaCl_2 \cdot 6H_2O$$ and $$MgCl_2 \cdot 8H_2O$$ undergo dehydration on heating.
Statement II: BeO is amphoteric, whereas the oxides of other elements in the same group are acidic.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 35
Among the following, the aromatic compounds are:
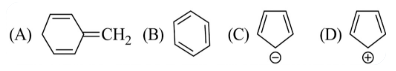
Choose the correct answer from the following options:
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 36
Assertion A: Enol form of acetone [$$CH_3COCH_3$$] exists in < 0.1% quantity. However, the enol form of acetyl acetone [$$CH_3COCH_2OCCH_3$$] exists in approximately 15% quantity.
Reason R: enol form of acetyl acetone is stabilized by intramolecular hydrogen bonding, which is not possible in enol form of acetone.
Choose the correct statement:
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 37
In chromatography technique, the purification of a compound is independent of:
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 38
The product "A" and "B" formed in below reactions are:
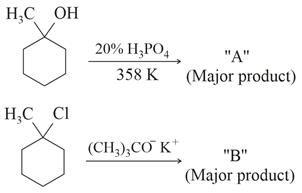
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 39
The type of pollution that gets increased during the daytime and in the presence of $$O_3$$ is:
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 40
Given below are two statements:
Statement I: The E° value of $$Ce^{4+}/Ce^{3+}$$ is +1.74 V.
Statement II: Ce is more stable in $$Ce^{4+}$$ state than $$Ce^{3+}$$ state.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 41
The process that involves the removal of sulphur from the ores is:
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 42
Match List-I with List-II:

Choose the correct answer from the options given below:
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 43
Match List-I with List-II:

Choose the correct answer from the options given below:
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 44
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: Size of $$Bk^{3+}$$ ion is less than $$Np^{3+}$$ ion.
Reason R: The above is a consequence of the lanthanide contraction.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 45
Which of the following is Lindlar catalyst?
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 46
The product "P" in the above reaction is:
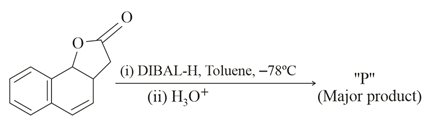
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 47
In the above chemical reaction, intermediate "X" and reagent/condition "A" are:
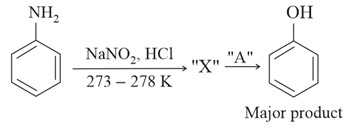
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 48
Which of the following reaction DOES NOT involve Hoffmann Bromamide degradation?
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 49
The functions of antihistamine are:
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 50
Which among the following pairs of Vitamins is stored in our body relatively for longer duration?
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 51
Complete combustion of 750 g of an organic compound provides 420 g of $$CO_2$$ and 210 g of $$H_2O$$. The percentage composition of carbon and hydrogen in organic compound is 15.3 and ________ respectively. (Round off to the Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 52
A 6.50 molal solution of KOH (aq.) has a density of 1.89 g cm$$^{-3}$$. The molarity of the solution is ________ mol dm$$^{-3}$$. (Round off to the Nearest Integer).
[Atomic masses: K: 39.0 u; O: 16.0 u; H: 1.0 u]
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 53
When light of wavelength 248 nm falls on a metal of threshold energy 3.0 eV, the de-Broglie wavelength of emitted electrons is ________ $$\mathring{A}$$ (angstrom). (Round off to the Nearest Integer).
[Use: $$\sqrt{3} = 1.73$$, $$h = 6.63 \times 10^{-34}$$ Js; $$m_e = 9.1 \times 10^{-31}$$ kg; $$c = 3.0 \times 10^{8}$$ ms$$^{-1}$$; $$1$$ eV $$= 1.6 \times 10^{-19}$$ J]
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 54
For the reaction $$A(g) \rightleftharpoons B(g)$$ at 495 K, $$\Delta_rG^\circ = -9.478$$ kJ mol$$^{-1}$$. If we start the reaction in a closed container at 495 K with 22 millimoles of A, the amount of B is the equilibrium mixture is ________ millimoles. (Round off to the Nearest Integer).
[$$R = 8.314$$ J mol$$^{-1}$$ K$$^{-1}$$; $$\ln 10 = 2.303$$]
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 55
Two salts $$A_2X$$ and MX have the same value of solubility product of $$4.0 \times 10^{-12}$$. The ratio of their molar solubilities i.e. $$\frac{S(A_2X)}{S(MX)}$$ = ________. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 56
$$2MnO_4^- + bC_2O_4^{2-} + cH^+ \rightarrow xMn^{2+} + yCO_2 + zH_2O$$
If the above equation is balanced with integer coefficients, the value of c is ________. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 57
A certain element crystallises in a bcc lattice of unit cell edge length 27 $$\mathring{A}$$. If the same element under the same conditions crystallises in the fcc lattice, the edge length of the unit cell in $$\mathring{A}$$ will be ________. (Round off to the Nearest Integer).
[Assume each lattice point has a single atom]
[Assume $$\sqrt{3} = 1.73$$, $$\sqrt{2} = 1.41$$]
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 58
$$AB_2$$ is 10% dissociated in water to $$A^{2+}$$ and $$B^-$$. The boiling point of 10.0 molal aqueous solution of $$AB_2$$ is ________ °C. (Round off to the Nearest Integer).
[Given: Molal elevation constant of water $$K_b = 0.5$$ K kg mol$$^{-1}$$, boiling point of pure water = 100°C]
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 59
The decomposition of formic acid on gold surface follows first order kinetics. If the rate constant at 300 K is $$1.0 \times 10^{-3}$$ s$$^{-1}$$ and the activation energy $$E_a = 11.488$$ kJ mol$$^{-1}$$, the rate constant at 200 K is ________ $$\times 10^{-5}$$ s$$^{-1}$$. (Round off to the Nearest Integer).
[Given $$R = 8.314$$ J mol$$^{-1}$$ K$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 16th March 2021 Shift 1 - Chemistry - Question 60
The equivalents of ethylene diamine required to replace the neutral ligands from the coordination sphere of the trans-complex of $$CoCl_3 \cdot 4NH_3$$ is ________. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




