NTA JEE Main 16 th April 2018 Online
For the following questions answer them individually
NTA JEE Main 16 th April 2018 Online - Question 41
A group 13 element 'X' reacts with chlorine gas to produce a compound XCl$$_3$$. XCl$$_3$$ is electron deficient and easily reacts with NH$$_3$$ to form Cl$$_3$$X $$\leftarrow$$ NH$$_3$$ adduct; however, XCl$$_3$$ does not dimerize. X is:
NTA JEE Main 16 th April 2018 Online - Question 42
The major product of the following reaction is:
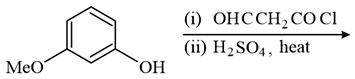
NTA JEE Main 16 th April 2018 Online - Question 43
The mass of a non-volatile, non-electrolyte solute (molar mass = 50 g mol$$^{-1}$$) needed to be dissolved in 114 g octane to reduce its vapour pressure by 75%, is:
NTA JEE Main 16 th April 2018 Online - Question 44
When 9.65 ampere current was passed for 1.0 hour into nitrobenzene in acidic medium, the amount of p-aminophenol produced is:
NTA JEE Main 16 th April 2018 Online - Question 45
If 50% of a reaction occurs in 100 second and 75% of the reaction occurs in 200 second, the order of this reaction is:
NTA JEE Main 16 th April 2018 Online - Question 46
Which one of the following is not a property of physical adsorption?
NTA JEE Main 16 th April 2018 Online - Question 47
In the extraction of copper from its sulphide ore, metal is finally obtained by the oxidation of cuprous sulphide with:
NTA JEE Main 16 th April 2018 Online - Question 48
Among the oxides of nitrogen: N$$_2$$O$$_3$$, N$$_2$$O$$_4$$ and N$$_2$$O$$_5$$; the molecule(s) having nitrogen-nitrogen bond is/are:
NTA JEE Main 16 th April 2018 Online - Question 49
When XO$$_2$$ is fused with an alkali metal hydroxide in presence of an oxidizing agent such as KNO$$_3$$; a dark green product is formed which disproportionate in acidic solution to afford a dark purple solution. X is:
NTA JEE Main 16 th April 2018 Online - Question 50
The incorrect statement is:


