NTA JEE Main 16 th April 2018 Online
For the following questions answer them individually
NTA JEE Main 16 th April 2018 Online - Question 1
Let $$\vec{A} = (\hat{i} + \hat{j})$$ and $$\vec{B} = (2\hat{i} - \hat{j})$$. The magnitude of a coplanar vector $$\vec{C}$$ such that $$\vec{A} \cdot \vec{C} = \vec{B} \cdot \vec{C} = \vec{A} \cdot \vec{B}$$ is given by:
NTA JEE Main 16 th April 2018 Online - Question 2
The percentage errors in quantities P, Q, R and S are 0.5%, 1%, 3% and 1.5% respectively in the measurement of a physical quantity $$A = \frac{P^3 Q^2}{\sqrt{RS}}$$. The maximum percentage error in the value of A will be:
NTA JEE Main 16 th April 2018 Online - Question 3
A body of mass m starts moving from rest along x-axis so that its velocity varies as $$v = a\sqrt{s}$$ where a is a constant and s is the distance covered by the body. The total work done by all the forces acting on the body in the first t second after the start of the motion is:
NTA JEE Main 16 th April 2018 Online - Question 4
Two particles of the same mass m are moving in circular orbits because of force, given by $$F(r) = -\frac{16}{r} - r^3$$. The first particle is at a distance r = 1, and the second, at r = 4. The best estimate for the ratio of kinetic energies of the first and the second particle is closest to:
NTA JEE Main 16 th April 2018 Online - Question 5
A thin circular disk is in the xy plane as shown in the figure. The ratio of its moment of inertia about z and z' axes will be:
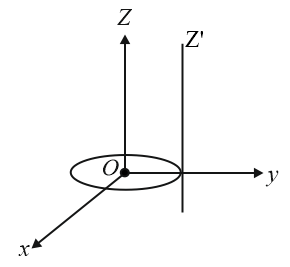
NTA JEE Main 16 th April 2018 Online - Question 6
The relative uncertainty in the period of a satellite orbiting around the earth is $$10^{-2}$$. If the relative uncertainty in the radius of the orbit is negligible, the relative uncertainty in the mass of the earth is:
NTA JEE Main 16 th April 2018 Online - Question 7
Suppose that the angular velocity of rotation of the Earth is increased. Then, as a consequence:
NTA JEE Main 16 th April 2018 Online - Question 8
A small soap bubble of radius 4 cm is trapped inside another bubble of radius 6 cm without any contact. Let P$$_2$$ be the pressure inside the inner bubble and P$$_0$$, the pressure outside the outer bubble. Radius of another bubble with pressure difference P$$_2$$ - P$$_0$$ between its inside and outside would be:
NTA JEE Main 16 th April 2018 Online - Question 9
One mole of an ideal monatomic gas is taken along the path ABCA as shown in the PV diagram. The maximum temperature attained by the gas along the path BC is given by:
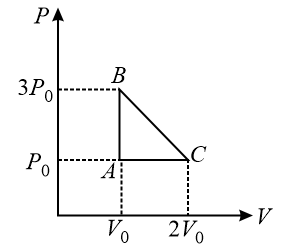
NTA JEE Main 16 th April 2018 Online - Question 10
Two moles of helium are mixed with n moles of hydrogen. If $$\frac{C_p}{C_v} = \frac{3}{2}$$ for the mixture then the value of n is:
.webp)


