NTA JEE Main 16 th April 2018 Online - Chemistry
For the following questions answer them individually
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 31
An unknown chlorohydrocarbon has 3.55% of chlorine. If each molecule of the hydrocarbon has one chlorine atom only; chlorine atoms present in 1 g of chlorohydrocarbon are: (Atomic wt. of Cl = 35.5 u; Avogadro constant = 6.023 $$\times$$ 10$$^{23}$$ mol$$^{-1}$$)
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 32
Which of the following statements is false?
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 33
Which of the following conversions involves change in both shape and hybridisation?
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 34
The most polar compound among the following is:
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 35
The incorrect geometry is represented by:
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 36
Assuming ideal gas behavior, the ratio of density of ammonia to that of hydrogen chloride at same temperature and pressure is: (Atomic weight of Cl is 35.5 u)
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 37
At 320 K, a gas A$$_2$$ is 20% dissociated to A(g). The standard Gibbs free energy change at 320 K and 1 atm in J mol$$^{-1}$$ is approximately: (R = 8.314 J K$$^{-1}$$ mol$$^{-1}$$; ln 2 = 0.693; ln 3 = 1.098)
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 38
For which of the following processes, $$\Delta S$$ is negative?
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 39
For standardizing NaOH solution, which of the following is used as a primary standard?
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 40
The gas phase reaction 2NO$$_2$$(g) $$\rightarrow$$ N$$_2$$O$$_4$$(g) is an exothermic reaction. The decomposition of N$$_2$$O$$_4$$, in equilibrium mixture of NO$$_2$$(g) and N$$_2$$O$$_4$$(g) can be increased by:
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 41
A group 13 element 'X' reacts with chlorine gas to produce a compound XCl$$_3$$. XCl$$_3$$ is electron deficient and easily reacts with NH$$_3$$ to form Cl$$_3$$X $$\leftarrow$$ NH$$_3$$ adduct; however, XCl$$_3$$ does not dimerize. X is:
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 42
The major product of the following reaction is:
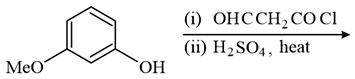
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 43
The mass of a non-volatile, non-electrolyte solute (molar mass = 50 g mol$$^{-1}$$) needed to be dissolved in 114 g octane to reduce its vapour pressure by 75%, is:
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 44
When 9.65 ampere current was passed for 1.0 hour into nitrobenzene in acidic medium, the amount of p-aminophenol produced is:
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 45
If 50% of a reaction occurs in 100 second and 75% of the reaction occurs in 200 second, the order of this reaction is:
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 46
Which one of the following is not a property of physical adsorption?
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 47
In the extraction of copper from its sulphide ore, metal is finally obtained by the oxidation of cuprous sulphide with:
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 48
Among the oxides of nitrogen: N$$_2$$O$$_3$$, N$$_2$$O$$_4$$ and N$$_2$$O$$_5$$; the molecule(s) having nitrogen-nitrogen bond is/are:
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 49
When XO$$_2$$ is fused with an alkali metal hydroxide in presence of an oxidizing agent such as KNO$$_3$$; a dark green product is formed which disproportionate in acidic solution to afford a dark purple solution. X is:
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 50
The incorrect statement is:
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 51
In a complexometric titration of metal ion with ligand M (Metal ion) + L(Ligand) $$\rightarrow$$ C (Complex). End point is estimated spectrophotometrically (through light absorption). If 'M' and 'C' do not absorb light and only 'L' absorbs then the titration plot between absorbed light (A) versus volume of ligand 'L' (V) would look like:
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 52
In Wilkinson's catalyst, the hybridization of central metal ion and its shape are respectively:
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 53
Which of the following complexes will show geometrical isomerism?
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 54
The major product of the following reaction is:
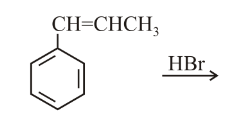
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 55
The major product of the following reaction is:
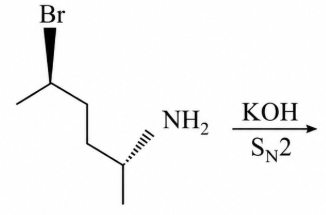
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 56
Which of the following compounds will most readily be dehydrated to give alkene under acidic condition?
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 57
The major product B formed in the following reaction sequence is:
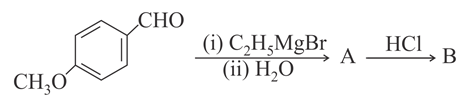
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 58
Products A and B formed in the following reactions are respectively:
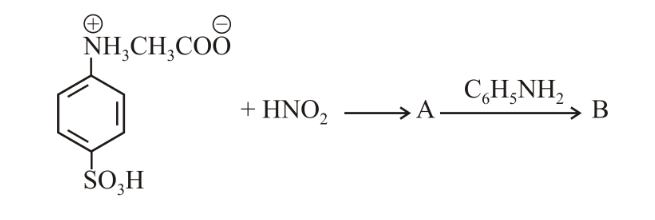
NTA JEE Main 16 th April 2018 Online - Chemistry - Question 59
The correct match between items of List-I and List-II is:

NTA JEE Main 16 th April 2018 Online - Chemistry - Question 60
Among the following, the incorrect statement is:
.webp)
.webp)
.webp)
.webp)




