NTA JEE Main 15th April 2023 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 31
Given below are two statements
Statement I : According to Bohr's model of hydrogen atom, the angular momentum of an electron in a given stationary state is quantised.
Statement II : The concept of electron in Bohr's orbit, violates the Heisenberg uncertainty principle. In the light of the above statements, choose the most appropriate answer from the options given below
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 32
Consider the following statement
(A) NF$$_3$$ molecules has a trigonal planar structure.
(B) Bond Length of N$$_2$$ is shorter than O$$_2$$.
(C) Isoelectronic molecules or ions have identical bond order.
(D) Dipole moment of H$$_2$$S is higher than that of water molecule.
Choose the correct answer from the options given below:
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 33
Which of the following statement(s) is/are correct?
(A) The pH of $$1 \times 10^{-8}$$ M HCl solution is 8.
(B) The conjugate base of H$$_2$$PO$$_4^-$$ is HPO$$_4^{2-}$$.
(C) K$$_w$$ increases with increase in temperature.
(D) When a solution of a weak monoprotic acid is titrated against a strong base at half neutralisation point, pH $$= \frac{1}{2}$$pK$$_a$$. Choose the correct answer from the options given below:
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 34
During water-gas shift reaction
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 35
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R:
Assertion (A) : BeCl$$_2$$ and MgCl$$_2$$ produce characteristic flame
Reason (R) : The excitation energy is high in BeCl$$_2$$ and MgCl$$_2$$
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 36
For a good quality cement, the ratio of silica to alumina is found to be
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 37
Which of the following statement is correct for paper chromatography?
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 38
Decreasing order of reactivity towards electrophilic substitution for the following compounds is:
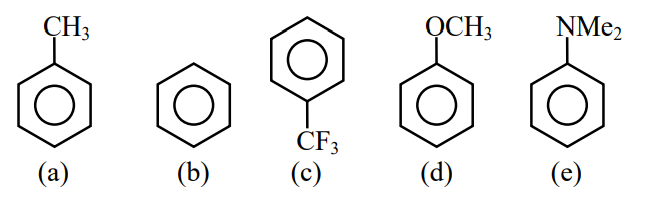
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 39
The product formed in the following multistep reaction is:
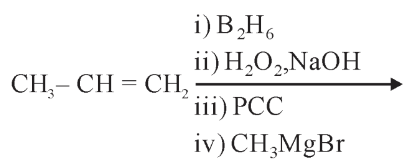
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 40
The possibility of photochemical smog formation will be minimum at
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 41
Which of the following expressions is correct in case of a CsCl unit cell (edge length 'a')?
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 42
Which one of the following is not an example of calcination?
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 43
The number of P $$-$$ O $$-$$ P bonds in H$$_4$$P$$_2$$O$$_7$$, (HPO$$_3$$)$$_3$$, and P$$_4$$O$$_{10}$$ are respectively
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 44
The complex with highest magnitude of crystal field splitting energy $$(\Delta_0)$$ is
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 45
The major product formed in the Friedel-Craft acylation of chlorobenzene is
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 46
A cyclic anhydride reacts with H$$_2$$O to give 'A' (major product).
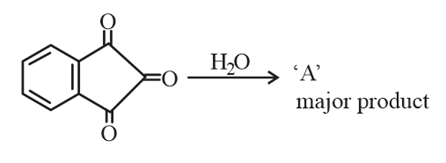
'A' formed in the above reaction is
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 47
Consider the following sequence of reactions
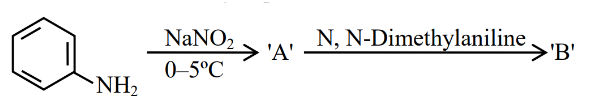
The product 'B' is
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 48
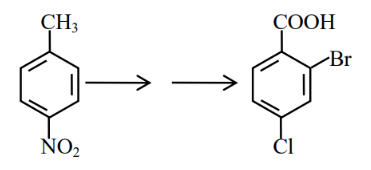
In the above conversion the correct sequence of reagents to be added is
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 49
Match List I with List II:
| List I (Monomer) | List II (Polymer) | ||
|---|---|---|---|
| A | Tetrafluoroethene | I | Orlon |
| B | Acrylonitrile | II | Natural rubber |
| C | Caprolactam | III | Teflon |
| D | Isoprene | IV | Nylon-6 |
Choose the correct answer from the options given below:
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 50
Which is not true for arginine?
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 51
The total number of isoelectronic species from the given set is _____.
O$$^{2-}$$, F$$^-$$, Al, Mg$$^{2+}$$, Na$$^+$$, O$$^+$$, Mg, Al$$^{3+}$$, F
789
456
123
0.-
Clear All
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 52
$$30.4$$ kJ of heat is required to melt one mole of sodium chloride and the entropy change at the melting point is $$28.4$$ J K$$^{-1}$$ mol$$^{-1}$$ at 1 atm. The melting point of sodium chloride is _____ K (Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 53
The total change in the oxidation state of manganese involved in the reaction of KMnO$$_4$$ and potassium iodide in the acidic medium is _____.
789
456
123
0.-
Clear All
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 54
The vapour pressure of $$30\%$$ (w/v), aqueous solution of glucose is _____ mm Hg at $$25°$$C.
[Given: The density of 30% (w/v), aqueous solution of glucose is $$1.2$$ g cm$$^{-3}$$ and vapour pressure of pure water is $$24$$ mm Hg.]
(Molar mass of glucose is 180 g mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 55
The number of correct statements from the following is _____
(A) Conductivity always decreases with decrease in concentration for both strong and weak electrolytes.
(B) The number of ions per unit volume that carry current in a solution increases on dilution.
(C) Molar conductivity increases with decrease in concentration.
(D) The variation in molar conductivity is different for strong and weak electrolytes.
(E) For weak electrolytes, the change in molar conductivity with dilution is due to decrease in degree of dissociation.
789
456
123
0.-
Clear All
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 56
For a reversible reaction A $$\rightleftharpoons$$ B, the $$\Delta H$$ forward reaction $$= 20$$ kJ mol$$^{-1}$$. The activation energy of the uncatalyzed forward reaction is $$300$$ kJ mol$$^{-1}$$. When the reaction is catalysed keeping the reactant concentration same, the rate of the catalysed forward reaction at $$27°$$C is found to be same as that of the uncatalyzed reaction at $$327°$$C. The activation energy of the catalysed backward reaction is _____ kJ mol$$^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 57
$$20$$ mL of $$0.5$$ M NaCl is required to coagulate $$200$$ mL of As$$_2$$S$$_3$$ solution in 2 hours. The coagulating value of NaCl is _____.
789
456
123
0.-
Clear All
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 58
In Chromyl chloride, the oxidation state of chromium is $$(+)$$ _____.
789
456
123
0.-
Clear All
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 59
The volume (in mL) of $$0.1$$ M AgNO$$_3$$ required for complete precipitation of chloride ions present in $$20$$ mL of $$0.01$$ M solution of [Cr(H$$_2$$O)$$_5$$Cl]Cl$$_2$$ as silver chloride is _____.
789
456
123
0.-
Clear All
NTA JEE Main 15th April 2023 Shift 1 - Chemistry - Question 60
The homoleptic and octahedral complex of Co$$^{2+}$$ and H$$_2$$O has _____ unpaired electron(s) in the t$$_{2g}$$ set of orbitals.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




