NTA JEE Main 15th April 2018 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 31
For per gram of reactant, the maximum quantity of N$$_2$$ gas is produced in which of the following thermal decomposition reactions? (Given: Atomic wt. : Cr = 52u, Ba = 137u)
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 32
The de-Broglie's wavelength of electron present in first Bohr orbit of 'H' atom is:
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 33
The correct order of electron affinity is:
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 34
Which of the following best describes the diagram of molecular orbital?
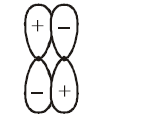
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 35
$$\Delta_r G^\circ$$ at 500 K for substance 'S' in liquid state and gaseous state are +100.7 kcal mol$$^{-1}$$ and +103 kcal mol$$^{-1}$$, respectively. Vapour pressure of liquid 'S' at 500 K is approximately equal to: (R = 2 cal K$$^{-1}$$ mol$$^{-1}$$)
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 36
Given (i) 2Fe$$_2$$O$$_3$$(s) $$\rightarrow$$ 4Fe(s) + 3O$$_2$$(g); $$\Delta_r G^\circ$$ = +1487.0 kJ mol$$^{-1}$$
(ii) 2CO(g) + O$$_2$$(g) $$\rightarrow$$ 2CO$$_2$$(g); $$\Delta_r G^\circ$$ = -514.4 kJ mol$$^{-1}$$
Free energy change, $$\Delta_r G^\circ$$ for the reaction 2Fe$$_2$$O$$_3$$(s) + 6CO(g) $$\rightarrow$$ 4Fe(s) + 6CO$$_2$$(g) will be:
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 37
At a certain temperature in a 5L vessel, 2 moles of carbon monoxide and 3 moles of chlorine were allowed to reach equilibrium according to the reaction, CO + Cl$$_2$$ $$\rightleftharpoons$$ COCl$$_2$$. At equilibrium, if one mole of CO is present then equilibrium constant (K$$_c$$) for the reaction is:
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 38
Following four solutions are prepared by mixing different volumes of NaOH and HCl of different concentrations, pH of which one of them will be equal to 1?
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 39
In KO$$_2$$, the nature of oxygen species and the oxidation state of oxygen atom are, respectively:
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 40
Lithium aluminium hydride reacts with silicon tetrachloride to form:
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 41
The total number of optically active compounds formed in the following reaction is:
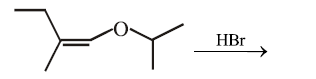
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 42
On the treatment of the following compound with a strong acid, the most susceptible site for bond cleavage is:
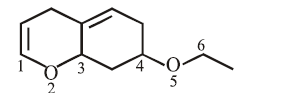
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 43
Two compounds I and II are eluted by column chromatography (adsorption of I > II). Which one of the following is a correct statement?
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 44
When 2-butyne is treated with H$$_2$$/Lindlar's catalyst, compound X is produced as the major product and when treated with Na/liq. NH$$_3$$ it produces Y as the major product. Which of the following statements is correct?
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 45
The major product formed in the following reaction is:
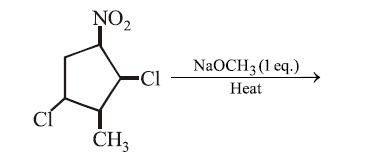
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 46
Biochemical oxygen demand (BOD) value can be a measure of water pollution caused by the organic matter. Which of the following statements is correct?
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 47
All of the following share the same crystal structure except:
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 48
Two 5 molal solutions are prepared by dissolving a non-electrolyte, non-volatile solute separately in the solvents X and Y. The molecular weights of the solvents are M$$_X$$ and M$$_Y$$, respectively where M$$_X$$ = $$\frac{3}{4}$$ M$$_Y$$. The relative lowering of vapour pressure of the solution in X is "m" times that of the solution in Y. Given that the number of moles of solute is very small in comparison to that of solvent, the value of "m" is:
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 49
For a first order reaction, A $$\rightarrow$$ P, t$$_{1/2}$$ (half-life) is 10 days. The time required for $$\frac{1}{4}$$th conversion of A (in days) is: (ln 2 = 0.693, ln 3 = 1.1)
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 50
If x gram of gas is adsorbed by m gram of adsorbent at pressure P the plot of log $$\frac{x}{m}$$ versus log P is linear. The slope of the plot is: (m and k are constants and n > 1)
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 51
In the leaching method, bauxite ore is digested with a concentrated solution of NaOH that produces 'X'. When CO$$_2$$ gas is passed through the aqueous solution of 'X', a hydrated compound 'Y' is precipitated. 'X' and 'Y' respectively are:
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 52
In XeO$$_3$$ F$$_2$$, the number of bond pair(s), $$\pi$$-bond(s) and lone pair(s) on Xe atom respectively are:
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 53
The number of P - O bonds in P$$_4$$O$$_6$$ is:
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 54
The correct order of spin-only magnetic moments among the following is: (Atomic number: Mn = 25, Co = 27, Ni = 28, Zn = 30)
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 55
The total number of possible isomers for square planar [Pt(Cl)(NO$$_2$$)(NO$$_3$$)(SCN)]$$^{2-}$$ is:
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 56
The major product formed in the following reaction is:
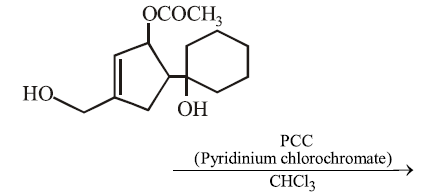
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 57
The increasing order of the acidity of the following carboxylic acids is:
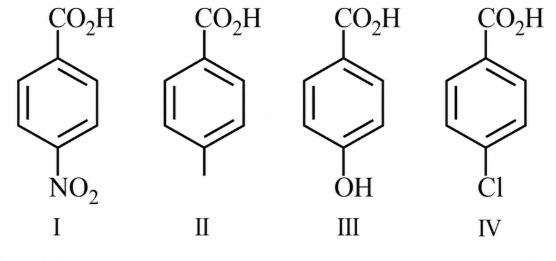
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 58
The increasing order of diazotisation of the following compounds is:
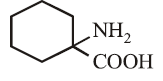
(a)
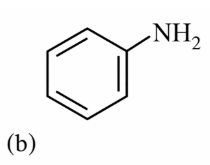
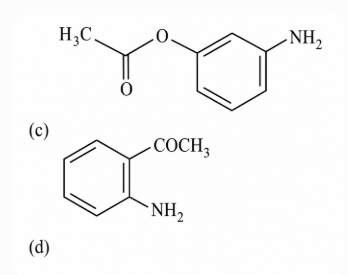
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 59
Which of the following statements is not true?
NTA JEE Main 15th April 2018 Shift 2 - Chemistry - Question 60
The dipeptide, Gln-Gly, on treatment with CH$$_3$$COCl followed by aqueous work up gives:
.webp)
.webp)
.webp)
.webp)




