NTA JEE Main 15th April 2018 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 31
A sample of NaClO$$_3$$ is converted by heat to NaCl with a loss of 0.16 g of oxygen. The residue is dissolved in water and precipitated as AgCl. The mass of AgCl (in g) obtained will be: (Given: Molar mass of AgCl = 143.5 g mol$$^{-1}$$)
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 32
Ejection of the photoelectron from metal in the photoelectric effect experiment can be stopped by applying 0.5 V when the radiation of 250 nm is used. The work function of the metal is:
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 33
For Na$$^+$$, Mg$$^{2+}$$, F$$^-$$ and O$$^{2-}$$; the correct order of increasing ionic radii is:
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 34
In the molecular orbital diagram for the molecular ion, N$$_2^+$$, the number of electrons in the $$\sigma_{2p}$$ molecular orbital is:
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 35
Identify the pair in which the geometry of the species is T-shape and square pyramidal, respectively:
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 36
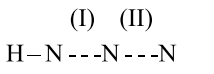
In hydrogen azide, the bond orders of bonds (I) and (II) are:
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 37
In graphite and diamond, the percentage of p characters of the hybrid orbitals in hybridization are respectively:
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 38
The decreasing order of bond angles in BF$$_3$$, NH$$_3$$, PF$$_3$$ and I$$_3^-$$ is:
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 39
For which of the following reactions, $$\Delta H$$ is equal to $$\Delta U$$?
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 40
An ideal gas undergoes a cyclic process as shown in Figure.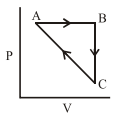
$$\Delta U_{BC} = -5$$ kJ mol$$^{-1}$$, $$q_{AB} = 2$$ kJ mol$$^{-1}$$
$$W_{AB} = -5$$ kJ mol$$^{-1}$$, $$W_{CA} = 3$$ kJ mol$$^{-1}$$
Heat absorbed by the system during process CA is:
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 41
In which of the following reactions, an increase in the volume of the container will favour the formation of products?
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 42
Which of the following is a Lewis acid?
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 43
The minimum volume of water required to dissolve 0.1 g lead (II) chloride to get a saturated solution ($$K_{SP}$$ of PbCl$$_2$$ = $$3.2 \times 10^{-8}$$; atomic mass of Pb = 207 u) is:
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 44
A white sodium salt dissolves readily in water to give a solution which is neutral to litmus. When silver nitrate solution is added to the aforementioned solution, a white precipitate is obtained which does not dissolve in dil. nitric acid. The anion is:
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 45
The correct match between items of List-I and List-II is:

NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 46
The IUPAC name of the following compound is: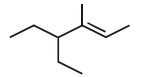
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 47
Which of the following will most readily give the dehydrohalogenation product?
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 48
The major product of the following reaction is: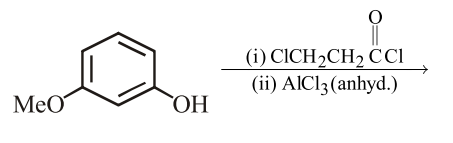
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 49
The increasing order of nitration of the following compounds is: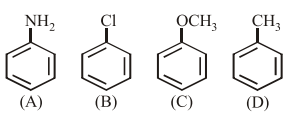
(A) Aniline, (B) Chlorobenzene, (C) Anisole, (D) Toluene
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 50
Which of the following arrangements shows the schematic alignment of magnetic moments of antiferromagnetic substance?
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 51
When an electric current is passes through acidified water, 112 mL of hydrogen gas at N.T.P. was collected at the cathode in 965 seconds. The current passed, in ampere, is:
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 52
N$$_2$$O$$_5$$ decomposes to NO$$_2$$ and O$$_2$$ and follows first order kinetics. After 50 minutes, the pressure inside the vessel increases from 50 mm Hg to 87.5 mm Hg. The pressure of the gaseous mixture after 100 minutes at constant temperature will be:
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 53
Which of the following statements about colloids is false?
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 54
Xenon hexafluoride on partial hydrolysis produces compounds 'X' and 'Y'. Compounds 'X' and 'Y' and the oxidation state of Xe are respectively:
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 55
The correct combination is:
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 56
The main reduction product of the following compound with NaBH$$_4$$ in methanol is: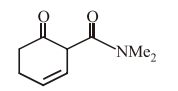
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 57
The reagent(s) required for the following conversion are: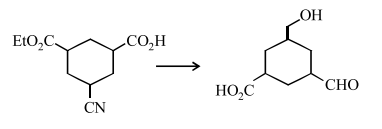
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 58
The copolymer formed by addition polymerization of styrene and acrylonitrile in the presence of peroxide is:
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 59
Which of the following will not exist in zwitter ionic form at pH = 7?
NTA JEE Main 15th April 2018 Shift 1 - Chemistry - Question 60
Which of the following is the correct structure of adenosine?
.webp)
.webp)
.webp)
.webp)




