NTA JEE Main 12th April 2023 Shift 1
For the following questions answer them individually
NTA JEE Main 12th April 2023 Shift 1 - Question 51
Values of work function ($$W_0$$) for a few metals are given below
| Metal | Li | Na | K | Mg | Cu | Ag |
|---|---|---|---|---|---|---|
| $$\frac{W_0}{eV}$$ | 2.42 | 2.3 | 2.25 | 3.7 | 4.8 | 4.3 |
The number of metals which will show photoelectric effect when light of wavelength $$400$$ nm falls on it is _____.
Given: $$h = 6.6 \times 10^{-34}$$ J s, $$c = 3 \times 10^8$$ ms$$^{-1}$$, $$e = 1.6 \times 10^{-19}$$ C
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Question 52
At $$600$$ K, the root mean square (rms) speed of gas X (molar mass $$= 40$$) is equal to the most probable speed of gas Y at $$90$$ K. The molar mass of the gas Y is _____ g mol$$^{-1}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Question 53
One mole of an ideal gas at $$350$$ K is in a $$2.0$$ L vessel of thermally conducting walls, which are in contact with the surroundings. It undergoes isothermal reversible expansion from $$2.0$$ L to $$3.0$$ L against a constant pressure of $$4$$ atm. The change in entropy of the surroundings ($$\Delta S$$) is _____ J K$$^{-1}$$ (Nearest integer)
Given: $$R = 8.314$$ J K$$^{-1}$$ mol$$^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Question 54
An analyst wants to convert $$1$$ L HCl of pH $$= 1$$ to a solution of HCl of pH $$= 2$$. The volume of water needed to do this dilution is _____ mL. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Question 55
Three organic compounds A, B and C were allowed to run in thin layer chromatography using hexane and gave the following result (see figure). The R$$_f$$ value of the most polar compound is _____ $$\times 10^{-2}$$
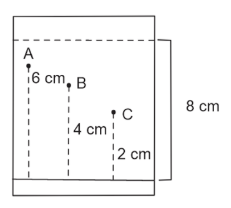
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Question 56
80 mole percent of MgCl$$_2$$ is dissociated in aqueous solution. The vapour pressure of $$1.0$$ molal aqueous solution of MgCl$$_2$$ at $$38°$$C is _____ mm Hg. (Nearest integer)
Given: Vapour pressure of water at $$38°$$C is $$50$$ mm Hg
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Question 57
The reaction $$2$$NO $$+ $$ Br$$_2 \to 2$$ NOBr takes place through the mechanism given below
NO $$+$$ Br$$_2 \rightleftharpoons$$ NOBr$$_2$$ (fast)
NOBr$$_2 +$$ NO $$\to 2$$ NOBr (slow)
The overall order of the reaction is _____.
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Question 58
Consider the following reaction sequence:
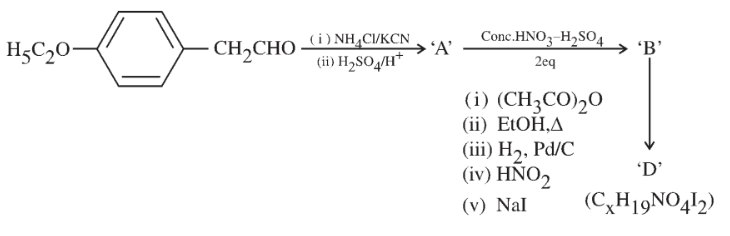
The value of x in compound 'D' is _____
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Question 59
The mass of NH$$_3$$ produced when $$131.8$$ kg of cyclohexane carbaldehyde undergoes Tollen's test is _____ kg. (Nearest Integer)
Molar mass of C $$= 12$$ g/mol, N $$= 14$$ g/mol, O $$= 16$$ g/mol
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Question 60
In an oligopeptide named Alanylglycylphenyl alanyl isoleucine, the number of sp$$^2$$ hybridised carbons is _____.
789
456
123
0.-
Clear All


