NTA JEE Main 12th April 2023 Shift 1
For the following questions answer them individually
NTA JEE Main 12th April 2023 Shift 1 - Question 31
A metal chloride contains $$55.0\%$$ of chlorine by weight. $$100$$ mL vapours of the metal chloride at STP weigh $$0.57$$ g. The molecular formula of the metal chloride is
(Given: Atomic mass of chlorine is $$35.5$$ u)
NTA JEE Main 12th April 2023 Shift 1 - Question 32
Given below are two statement : one is labelled as Assertion A and the other is labelled as Reason R
Assertion A : 5f electron can participate in bonding to a far greater extent than 4f electrons
Reason R : 5f orbitals are not as buried as 4f orbitals
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 12th April 2023 Shift 1 - Question 33
The bond order and magnetic property of acetylide ion are same as that of
NTA JEE Main 12th April 2023 Shift 1 - Question 34
Given below are two statements:
Statement I: SbCl$$_5$$ is more covalent than SbCl$$_3$$
Statement II: The higher oxides of halogens also tend to be more stable than the lower ones.
In the light of the above statements, choose the most appropriate answer from the options given below.
NTA JEE Main 12th April 2023 Shift 1 - Question 35
Match List I with List II
| List I Type of Hydride | List II Example | ||
|---|---|---|---|
| A | Electron deficient hydride | I | MgH$$_2$$ |
| B | Electron rich hydride | II | HF |
| C | Electron precise hydride | III | B$$_2$$H$$_6$$ |
| D | Saline hydride | IV | CH$$_4$$ |
Choose the correct answer from the options given below :
NTA JEE Main 12th April 2023 Shift 1 - Question 36
In the given reaction cycle
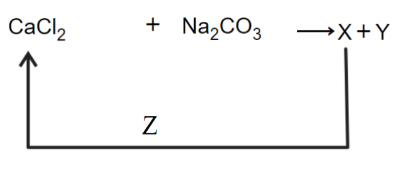
X, Y and Z respectively are
NTA JEE Main 12th April 2023 Shift 1 - Question 37
The density of alkali metals is in the order
NTA JEE Main 12th April 2023 Shift 1 - Question 38
Given below are two statements:
Statement I: Boron is extremely hard indicating its high lattice energy.
Statement II: Boron has highest melting and boiling point compared to its other group members.
In the light of the above statements, choose the most appropriate answer from the options given below
NTA JEE Main 12th April 2023 Shift 1 - Question 39
Correct statements for the given reaction are:
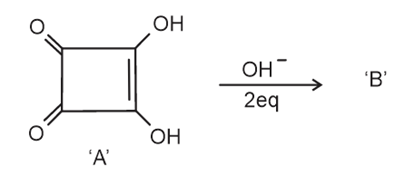
A. Compound 'B' is aromatic
B. The completion of above reaction is very slow
C. 'A' shows tautomerism
D. The bond lengths of C $$-$$ C in compound B are found to be same
Choose the correct answer from the options given below.
NTA JEE Main 12th April 2023 Shift 1 - Question 40
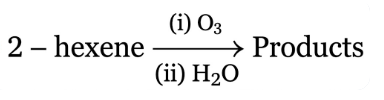
The two products formed in above reaction are


