NTA JEE Main 12th April 2023 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 31
A metal chloride contains $$55.0\%$$ of chlorine by weight. $$100$$ mL vapours of the metal chloride at STP weigh $$0.57$$ g. The molecular formula of the metal chloride is
(Given: Atomic mass of chlorine is $$35.5$$ u)
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 32
Given below are two statement : one is labelled as Assertion A and the other is labelled as Reason R
Assertion A : 5f electron can participate in bonding to a far greater extent than 4f electrons
Reason R : 5f orbitals are not as buried as 4f orbitals
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 33
The bond order and magnetic property of acetylide ion are same as that of
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 34
Given below are two statements:
Statement I: SbCl$$_5$$ is more covalent than SbCl$$_3$$
Statement II: The higher oxides of halogens also tend to be more stable than the lower ones.
In the light of the above statements, choose the most appropriate answer from the options given below.
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 35
Match List I with List II
| List I Type of Hydride | List II Example | ||
|---|---|---|---|
| A | Electron deficient hydride | I | MgH$$_2$$ |
| B | Electron rich hydride | II | HF |
| C | Electron precise hydride | III | B$$_2$$H$$_6$$ |
| D | Saline hydride | IV | CH$$_4$$ |
Choose the correct answer from the options given below :
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 36
In the given reaction cycle
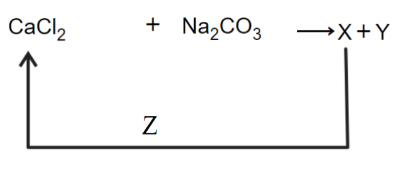
X, Y and Z respectively are
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 37
The density of alkali metals is in the order
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 38
Given below are two statements:
Statement I: Boron is extremely hard indicating its high lattice energy.
Statement II: Boron has highest melting and boiling point compared to its other group members.
In the light of the above statements, choose the most appropriate answer from the options given below
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 39
Correct statements for the given reaction are:
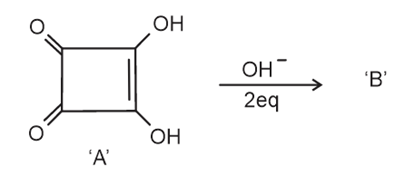
A. Compound 'B' is aromatic
B. The completion of above reaction is very slow
C. 'A' shows tautomerism
D. The bond lengths of C $$-$$ C in compound B are found to be same
Choose the correct answer from the options given below.
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 40
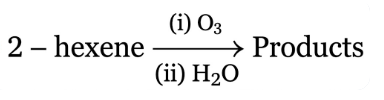
The two products formed in above reaction are
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 41
Match List I with List II
| List I | List II | ||
|---|---|---|---|
| A | Nitrogen oxides in air | I | Eutrophication |
| B | Methane in air | II | pH of rain water becomes 5.6 |
| C | Carbon dioxide | III | Global warming |
| D | Phosphate fertilisers in water | IV | Acid rain |
Choose the correct answer from the options given below :
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 42
For lead storage battery pick the correct statements
A. During charging of battery, PbSO$$_4$$ on anode is converted into PbO$$_2$$
B. During charging of battery, PbSO$$_4$$ on cathode is converted into PbO$$_2$$
C. Lead storage battery consists of grid of lead packed with PbO$$_2$$ as anode
D. Lead storage battery has ~38% solution of sulphuric acid as an electrolyte
Choose the correct answer from the options given below:
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 43
Four gases, A, B, C and D have critical temperatures 5.3, 33.2, 126.0 and 154.3K respectively. For their adsorption on a fixed amount of charcoal, the correct order is :
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 44
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: In the Ellingham diagram, a sharp change in slope of the line is observed from Mg $$\to$$ MgO at ~1120°C
Reason R: There is a large change of entropy associated with the change of state
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 45
The incorrect statement regarding the reaction given below is
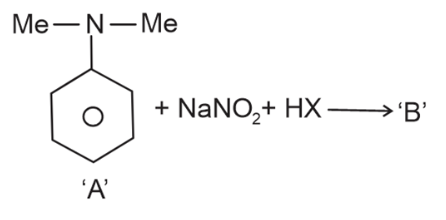
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 46
Match List I with List II
| List I Complex | List II CFSE ($$\Delta_0$$) | ||
|---|---|---|---|
| A | $$[Cu(NH_3)_6]^{2+}$$ | I | $$-0.6$$ |
| B | $$[Ti(H_2O)_6]^{3+}$$ | II | $$-2.0$$ |
| C | $$[Fe(CN)_6]^{3-}$$ | III | $$-1.2$$ |
| D | $$[NiF_6]^{4-}$$ | IV | $$-0.4$$ |
Choose the correct answer from the options given below:
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 47
In the following reaction
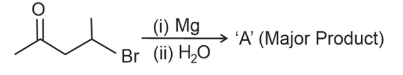
'A' (Major Product) is
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 48
The reaction of a diketone (MeCO-CH$$_2$$-CO-CH$$_3$$) with OEt$$^-$$/$$\Delta$$ gives 'A' (Major Product).
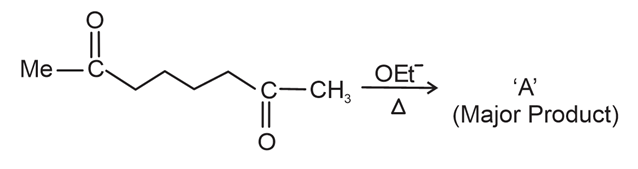
A in the above reaction is :
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 49
The major product 'P' formed in the following sequence of reactions is
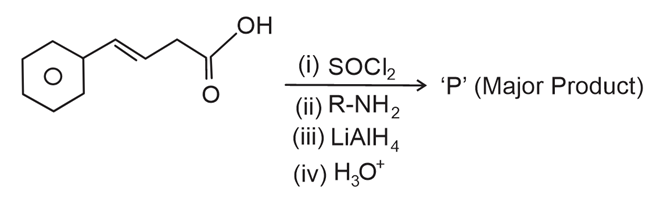
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 50
Match List I with List II
| List I (Example) | List II (Type) | ||
|---|---|---|---|
| A | 2-chloro-1, 3-butadiene | I | Biodegradable polymer |
| B | Nylon 2-nylon 6 | II | Synthetic Rubber |
| C | Polyacrylonitrile | III | Polyester |
| D | Dacron | IV | Addition Polymer |
Choose the correct answer from the options given below:
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 51
Values of work function ($$W_0$$) for a few metals are given below
| Metal | Li | Na | K | Mg | Cu | Ag |
|---|---|---|---|---|---|---|
| $$\frac{W_0}{eV}$$ | 2.42 | 2.3 | 2.25 | 3.7 | 4.8 | 4.3 |
The number of metals which will show photoelectric effect when light of wavelength $$400$$ nm falls on it is _____.
Given: $$h = 6.6 \times 10^{-34}$$ J s, $$c = 3 \times 10^8$$ ms$$^{-1}$$, $$e = 1.6 \times 10^{-19}$$ C
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 52
At $$600$$ K, the root mean square (rms) speed of gas X (molar mass $$= 40$$) is equal to the most probable speed of gas Y at $$90$$ K. The molar mass of the gas Y is _____ g mol$$^{-1}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 53
One mole of an ideal gas at $$350$$ K is in a $$2.0$$ L vessel of thermally conducting walls, which are in contact with the surroundings. It undergoes isothermal reversible expansion from $$2.0$$ L to $$3.0$$ L against a constant pressure of $$4$$ atm. The change in entropy of the surroundings ($$\Delta S$$) is _____ J K$$^{-1}$$ (Nearest integer)
Given: $$R = 8.314$$ J K$$^{-1}$$ mol$$^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 54
An analyst wants to convert $$1$$ L HCl of pH $$= 1$$ to a solution of HCl of pH $$= 2$$. The volume of water needed to do this dilution is _____ mL. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 55
Three organic compounds A, B and C were allowed to run in thin layer chromatography using hexane and gave the following result (see figure). The R$$_f$$ value of the most polar compound is _____ $$\times 10^{-2}$$
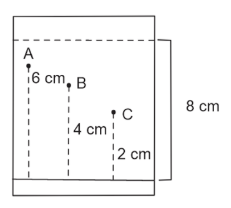
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 56
80 mole percent of MgCl$$_2$$ is dissociated in aqueous solution. The vapour pressure of $$1.0$$ molal aqueous solution of MgCl$$_2$$ at $$38°$$C is _____ mm Hg. (Nearest integer)
Given: Vapour pressure of water at $$38°$$C is $$50$$ mm Hg
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 57
The reaction $$2$$NO $$+ $$ Br$$_2 \to 2$$ NOBr takes place through the mechanism given below
NO $$+$$ Br$$_2 \rightleftharpoons$$ NOBr$$_2$$ (fast)
NOBr$$_2 +$$ NO $$\to 2$$ NOBr (slow)
The overall order of the reaction is _____.
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 58
Consider the following reaction sequence:
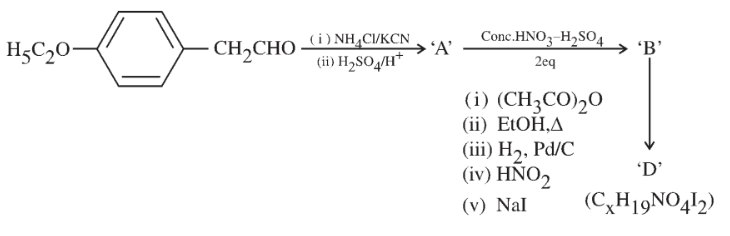
The value of x in compound 'D' is _____
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 59
The mass of NH$$_3$$ produced when $$131.8$$ kg of cyclohexane carbaldehyde undergoes Tollen's test is _____ kg. (Nearest Integer)
Molar mass of C $$= 12$$ g/mol, N $$= 14$$ g/mol, O $$= 16$$ g/mol
789
456
123
0.-
Clear All
NTA JEE Main 12th April 2023 Shift 1 - Chemistry - Question 60
In an oligopeptide named Alanylglycylphenyl alanyl isoleucine, the number of sp$$^2$$ hybridised carbons is _____.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




