NTA JEE Main 12th April 2014 Online
For the following questions answer them individually
NTA JEE Main 12th April 2014 Online - Question 41
The conjugate base of hydrazoic acid is:
NTA JEE Main 12th April 2014 Online - Question 42
Hydrogen peroxide acts both as an oxidising and as a reducing agent depending upon the nature of the reacting species. In which of the following cases H$$_2$$O$$_2$$ acts as a reducing agent in acid medium?
NTA JEE Main 12th April 2014 Online - Question 43
Which one of the following acids does not exhibit optical isomerism?
NTA JEE Main 12th April 2014 Online - Question 44
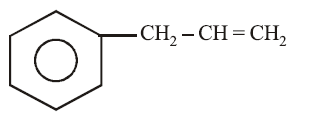
on mercuration-demercuration produces the major product:
NTA JEE Main 12th April 2014 Online - Question 45
In the presence of peroxide, HCl and HI do not give anti-Markownikoff's addition of alkenes because:
NTA JEE Main 12th April 2014 Online - Question 46
Global warming is due to increase of:
NTA JEE Main 12th April 2014 Online - Question 47
In a monoclinic unit cell, the relation of sides and angles are respectively:
NTA JEE Main 12th April 2014 Online - Question 48
How many electrons would be required to deposit 6.35 g of copper at the cathode during the electrolysis of an aqueous solution of copper sulphate? (Atomic mass of copper = 63.5u, N$$_A$$ = Avogadro's constant):
NTA JEE Main 12th April 2014 Online - Question 49
The rate coefficient (k) for a particular reactions is $$1.3 \times 10^{-4}$$ M$$^{-1}$$ s$$^{-1}$$ at 100°C, and $$1.3 \times 10^{-3}$$ M$$^{-1}$$ s$$^{-1}$$ at 150°C. What is the energy of activation (E$$_A$$) (in kJ) for this reaction? (R = molar gas constant = 8.314 JK$$^{-1}$$ mol$$^{-1}$$)
NTA JEE Main 12th April 2014 Online - Question 50
Which of the following xenon-oxo compounds may not be obtained by hydrolysis of xenon fluorides?


