NTA JEE Main 11th January 2019 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 31
25 mL of the given HCl solution requires 30 mL of 0.1M sodium carbonate solution. What is the volume of this HCl solution required to titrate 30 mL of 0.2M aqueous NaOH solution?
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 32
The de Broglie wavelength $$(\lambda)$$ associated with a photoelectron varies with the frequency $$(\nu)$$ of the incident radiation as, [$$\nu_0$$ is threshold frequency]:
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 33
The correct option with respect to the Pauling electronegativity values of the elements is:
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 34
The reaction $$MgO(s) + C(s) \to Mg(s) + CO(g)$$, for which $$\Delta H° = +491.1$$ kJ mol$$^{-1}$$ and $$\Delta S° = 198.0$$ JK$$^{-1}$$ mol$$^{-1}$$ is not feasible at 298 K. Temperature above which reaction will be feasible is
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 35
The standard reaction Gibbs energy for a chemical reaction at an absolute temperature T is given by $$\Delta G° = A - BT$$ where A and B are non-zero constants. Which of the following is true about this reaction?
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 36
For the equilibrium $$2H_2O \rightleftharpoons H_3O^+ + OH^-$$; the value of $$\Delta G°$$ at 298 K is approximately:
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 37
Match the following items in column I with the corresponding items in column II.

NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 38
The hydride that is NOT electron deficient is:
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 39
The relative stability of +1 oxidation state of group 13 elements follows the order
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 40
Which of the following compounds reacts with ethylmagnesium bromide and also decolourizes bromine water solution?
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 41
Which of the following compounds will produce a precipitate with $$AgNO_3$$?
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 42
Taj Mahal is being slowly disfigured and discoloured. This is primarily due to
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 43
The higher concentration of which gas in air can cause stiffness of flower buds?
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 44
The radius of the largest sphere which fits properly at the centre of the edge of a body centred cubic unit cell is: (Edge length is represented by 'a')
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 45
$$K_2HgI_4$$ is 40% ionised in aqueous solution. The value of its van't Hoff factor (i) is:
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 46
Given the equilibrium constant: $$K_C$$ of the reaction: $$Cu(s) + 2Ag^+(aq) \to Cu^{2+}(aq) + 2Ag(s)$$ is $$10 \times 10^{15}$$. Calculate the $$E°_{cell}$$ of this reaction at 298 K. [$$2.303 \frac{RT}{F}$$ at 298 K = 0.059 V]
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 47
The reaction $$2X \to B$$ is a zeroth order reaction. If the initial concentration of X is 0.2M, the half-life is 6 h. When the initial concentration of X is 0.5M, the time required to reach its final concentration of 0.2M will be
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 48
Among the colloids cheese (C), milk (M) and smoke (S), the correct combination of the dispersed phase and dispersion medium, respectively is:
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 49
The reaction that does NOT define calcination is:
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 50
$$A \xrightarrow{4KOH, O_2} 2B_{(Green)} + 2H_2O$$; $$B \xrightarrow{4HCl} 2C_{(Purple)} + MnO_2 + 2H_2O$$; $$2C \xrightarrow{H_2O, KI} 2A + KOH + D$$. In the above sequence of reactions, A and D, respectively, are:
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 51
The coordination number of Th in $$K_4[Th(C_2O_4)_4(H_2O)_2]$$ is: ($$C_2O_4^{2-}$$ = oxalato)
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 52
The number of bridging CO ligand(s) and Co-Co bond(s) in $$Co_2(CO)_8$$, respectively are:
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 53
The major product of the following reaction is:
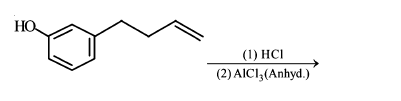
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 54
The major product obtained in the following conversion is:
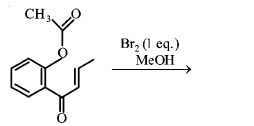
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 55
The major product obtained in the following reaction is:
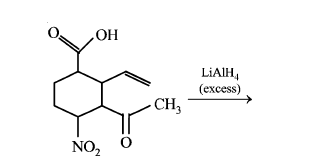
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 56
In the following compound, the favourable site/s for protonation is/are:
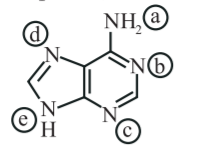
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 57
A compound 'X' on treatment with Br$$_2$$/NaOH, provided $$C_3H_9N$$, which gives positive carbylamine test. Compound X' is:
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 58
The homopolymer formed from 4-hydroxy-butanoic acids is:
NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 59
The correct match between Item I and Item II is:

NTA JEE Main 11th January 2019 Shift 2 - Chemistry - Question 60
The correct match between Item I and Item II is:

.webp)
.webp)
.webp)
.webp)




