NTA JEE Main 11th January 2019 Shift 1
For the following questions answer them individually
NTA JEE Main 11th January 2019 Shift 1 - Question 41
The correct match between items I and II is:

NTA JEE Main 11th January 2019 Shift 1 - Question 42
An organic compound is estimated through Dumas method and was found to evolve 6 moles of $$CO_2$$, 4 moles of $$H_2O$$ and 1 mole of nitrogen gas. The formula of the compound is:
NTA JEE Main 11th January 2019 Shift 1 - Question 43
Peroxyacetyl nitrate (PAN), an eye irritant, is produced by:
NTA JEE Main 11th January 2019 Shift 1 - Question 44
The concentration of dissolved oxygen (DO) in cold water can go upto
NTA JEE Main 11th January 2019 Shift 1 - Question 45
A solid having density of $$9 \times 10^3$$ kg m$$^{-3}$$ forms face centred cubic crystals of edge length $$200\sqrt{2}$$ pm. What is the molar mass of the solid? [Avogadro constant $$\approx 6 \times 10^{23}$$ mol$$^{-1}$$, $$\pi \approx 3$$]
NTA JEE Main 11th January 2019 Shift 1 - Question 46
The freezing point of a diluted milk sample is found to be $$-0.2°C$$, while it should have been $$-0.5°C$$ for pure milk. How much water has been added to pure milk to make the diluted sample?
NTA JEE Main 11th January 2019 Shift 1 - Question 47
For the cell $$Zn(s) | Zn^{2+}(aq) || M^{x+}(aq) | M(s)$$, different half cells and their standard electrode potentials are given below:
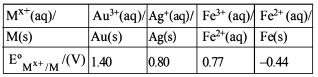
If $$E°_{Zn^{2+}/Zn} = -0.76$$ V, which cathode will give a maximum value of $$E°_{cell}$$ per electron transferred?
NTA JEE Main 11th January 2019 Shift 1 - Question 48
If a reaction follows the Arrhenius equation, the plot $$\ln k$$ vs $$\frac{1}{(RT)}$$ gives straight line with a gradient $$(-y)$$ unit. The energy required to activate the reactant is:
NTA JEE Main 11th January 2019 Shift 1 - Question 49
An example of solid sol is:
NTA JEE Main 11th January 2019 Shift 1 - Question 50
Match the ores (column A) with the metals (column B):



