NTA JEE Main 11th April 2023 Shift 2
For the following questions answer them individually
NTA JEE Main 11th April 2023 Shift 2 - Question 31
A solution is prepared by adding 2 g of 'X' to 1 mole of water. Mass percent of 'X' in solution is
NTA JEE Main 11th April 2023 Shift 2 - Question 32
Which one of the following pairs is an example of polar molecular solids?
NTA JEE Main 11th April 2023 Shift 2 - Question 33
Which hydride among the following is less stable?
NTA JEE Main 11th April 2023 Shift 2 - Question 34
Alkali metal from the following with least melting point is
NTA JEE Main 11th April 2023 Shift 2 - Question 35
Compound from the following that will not produce precipitate on reaction with AgNO$$_3$$ is
NTA JEE Main 11th April 2023 Shift 2 - Question 36
Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A:
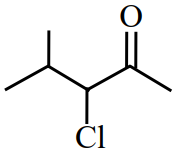
can be subjected to Wolff-Kishner reduction to give
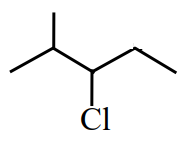
Reason R: Wolff-Kishner reduction is used to convert
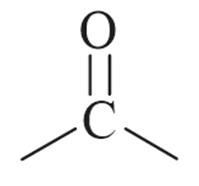
into
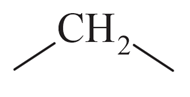
NTA JEE Main 11th April 2023 Shift 2 - Question 37
The major product formed in the following reaction is:
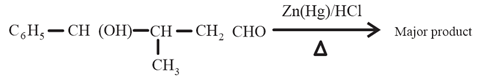
(a)
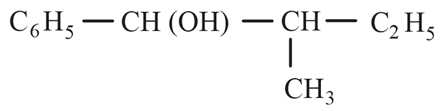
(b)
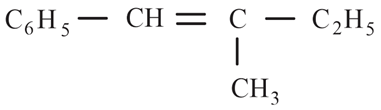
(c)
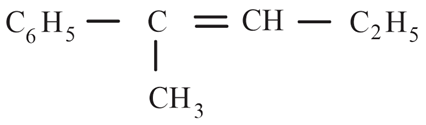
(d)
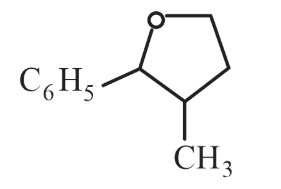
choose the correct answer from the options Given below:
NTA JEE Main 11th April 2023 Shift 2 - Question 38
Which of the following compounds is an example of Freon?
NTA JEE Main 11th April 2023 Shift 2 - Question 39
What weight of glucose must be dissolved in 100 g of water to lower the vapour pressure by 0.20 mm Hg?
(Assume dilute solution is being formed)
Given: Vapour pressure of pure water is 54.2 mm Hg at room temperature. Molar mass of glucose is 180 g mol$$^{-1}$$
NTA JEE Main 11th April 2023 Shift 2 - Question 40
For a chemical reaction $$A + B \to$$ Product, the order is 1 with respect to $$A$$ and $$B$$.
| Rate (mol L$$^{-1}$$ s$$^{-1}$$) | [A] (mol L$$^{-1}$$) | [B] (mol L$$^{-1}$$) |
| 0.10 | 20 | 0.5 |
| 0.40 | x | 0.5 |
| 0.80 | 40 | y |
What is the value of x and y?


