NTA JEE Main 11th April 2023 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 31
A solution is prepared by adding 2 g of 'X' to 1 mole of water. Mass percent of 'X' in solution is
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 32
Which one of the following pairs is an example of polar molecular solids?
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 33
Which hydride among the following is less stable?
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 34
Alkali metal from the following with least melting point is
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 35
Compound from the following that will not produce precipitate on reaction with AgNO$$_3$$ is
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 36
Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A:
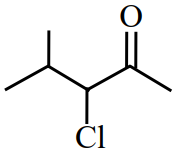
can be subjected to Wolff-Kishner reduction to give
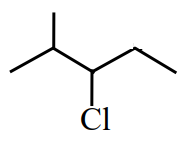
Reason R: Wolff-Kishner reduction is used to convert
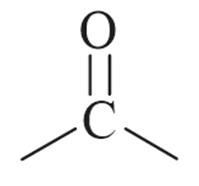
into
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 37
The major product formed in the following reaction is:
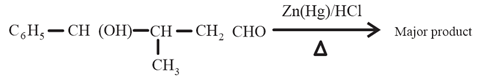
(a)
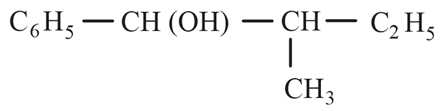
(b)
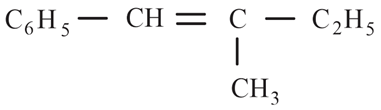
(c)
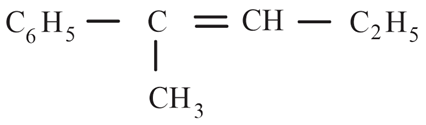
(d)
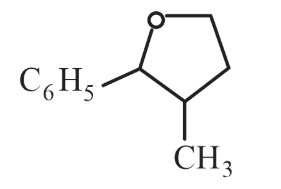
choose the correct answer from the options Given below:
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 38
Which of the following compounds is an example of Freon?
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 39
What weight of glucose must be dissolved in 100 g of water to lower the vapour pressure by 0.20 mm Hg?
(Assume dilute solution is being formed)
Given: Vapour pressure of pure water is 54.2 mm Hg at room temperature. Molar mass of glucose is 180 g mol$$^{-1}$$
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 40
For a chemical reaction $$A + B \to$$ Product, the order is 1 with respect to $$A$$ and $$B$$.
| Rate (mol L$$^{-1}$$ s$$^{-1}$$) | [A] (mol L$$^{-1}$$) | [B] (mol L$$^{-1}$$) |
| 0.10 | 20 | 0.5 |
| 0.40 | x | 0.5 |
| 0.80 | 40 | y |
What is the value of x and y?
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 41
Given below are two statements:
Statement I: In the metallurgy process, sulphide ore is converted to oxide before reduction.
Statement II: Oxide ores in general are easier to reduce.
In the light of the above statements, choose the most appropriate answer from the options below:
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 42
Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: A solution of the product obtained by heating a mole of glycine with a mole of chlorine in presence of red phosphorous generates chiral carbon atom.
Reason R: A molecule with 2 chiral carbons is always optically active.
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 43
One mole of P$$_4$$ reacts with 8 moles of SOCl$$_2$$ to give 4 moles of A, x mole of SO$$_2$$ and 2 moles of B. A, B and x respectively are
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 44
If Ni$$^{2+}$$ is replaced by Pt$$^{2+}$$ in the complex [NiCl$$_2$$Br$$_2$$]$$^{2-}$$, which of the following properties are expected to get changed?
A. Geometry
B. Geometrical isomerism
C. Optical isomerism
D. Magnetic properties
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 45
Match List I with List II
| LIST-I Complex | LIST-II Colour | ||
|---|---|---|---|
| A. | Mg(NH$$_4$$)PO$$_4$$ | I. | brown |
| B. | K$$_3$$[Co(NO$$_2$$)$$_6$$] | II. | white |
| C. | MnO(OH)$$_2$$ | III. | yellow |
| D. | Fe$$_4$$[Fe(CN)$$_6$$]$$_3$$ | IV. | blue |
Choose the correct answer from the options given below:
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 46
Given below are two statements, one is labelled as assertion A and the other is labelled as Reason R.
Assertion A: [CoCl(NH$$_3$$)$$_5$$]$$^{2+}$$ absorbs at lower wavelength of light with respect to [Co(NH$$_3$$)$$_5$$(H$$_2$$O)]$$^{3+}$$
Reason R: It is because the wavelength of light absorbed depends on the oxidation state of the metal ion.
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 47
The magnetic moment is measured in Bohr Magneton (BM). Spin only magnetic moment of Fe in [Fe(H$$_2$$O)$$_6$$]$$^{3+}$$ and [Fe(CN)$$_6$$]$$^{3-}$$ complexes respectively is:
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 48
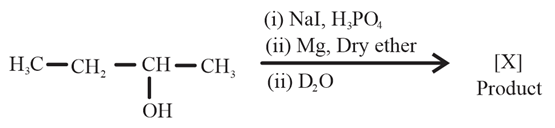
Product [X] formed in the above reaction is :
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 49
Compound 'B' is :
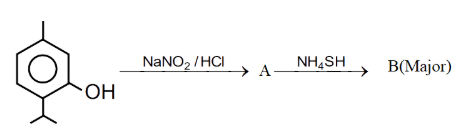
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 50
Given below are two statements:
Statement I: Ethane at 333 to 343 K and 6-7 atm pressure in the presence of AlEt$$_3$$ and TiCl$$_4$$ undergoes addition polymerization to give LDP.
Statement II: Caprolactam at 533-543 K in H$$_2$$O through step growth polymerizes to give Nylon 6.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 51
The volume of hydrogen liberated at STP by treating 2.4 g of magnesium with excess of hydrochloric acid is _______ $$\times 10^{-2}$$ L. Given Molar volume of gas is 22.4 L at STP. Molar mass of magnesium is 24 g mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 52
The number of correct statements from the following is _______
A. For 1s orbital, the probability density is maximum at the nucleus
B. For 2s orbital, the probability density first increases to maximum and then decreases sharply to zero.
C. Boundary surface diagrams of the orbitals encloses a region of 100% probability of finding the electron.
D. p and d-orbitals have 1 and 2 angular nodes respectively
E. probability density of p-orbital is zero at the nucleus
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 53
The maximum number of lone pairs of electron on the central atom from the following species is _______ ClO$$_3^-$$, XeF$$_4$$, SF$$_4$$ and I$$_3^-$$
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 54
The total number of intensive properties from the following is _______
Volume, Molar heat capacity, molarity, $$E^\circ_{cell}$$, Gibbs free energy change, Molar mass, Mole
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 55
4.5 moles each of hydrogen and iodine is heated in a sealed ten litre vessel. At equilibrium, 3 moles of HI were found. The equilibrium constant for $$H_2(g) + I_2(g) \rightleftharpoons 2HI_{(g)}$$ is _______
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 56
Mg(NO$$_3$$)$$_2$$.XH$$_2$$O and Ba(NO$$_3$$)$$_2$$.YH$$_2$$O, represent formula of the crystalline forms of nitrate salts. Sum of X and Y is _______
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 57
The number of possible isomeric products formed when 3-chloro-1-butene reacts with HCl through carbocation formation is _______
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 58
The number of correct statements from the following is _______
A. E$$_{cell}$$ is an intensive parameter
B. A negative E$$^\circ$$ means that the redox couple is a stronger reducing agent than the H$$^+$$/H$$_2$$ couple.
C. The amount of electricity required for oxidation or reduction depends on the stoichiometry of the electrode reaction.
D. The amount of chemical reaction which occurs at any electrode during electrolysis by a current is proportional to the quantity of electricity passed through the electrolyte.
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 59
The number of correct statements about modern adsorption theory of heterogeneous catalysis from the following is _______
A. The catalyst is diffused over the surface of reactants.
B. Reactants are adsorbed on the surface of the catalyst.
C. Occurrence of chemical reaction on the catalyst's surface through formation of an intermediate.
D. It is a combination of intermediate compound formation theory and the old adsorption theory.
E. It explains the action of the catalyst as well as those of catalytic promoters and poisons.
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 2 - Chemistry - Question 60
Number of compounds from the following which will not produce orange red precipitate with Benedict solution is _______
Glucose, maltose, sucrose, ribose, 2-deoxyribose, amylose, lactose
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




