NTA JEE Main 11th April 2023 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 31
25 mL of silver nitrate solution (1M) is added dropwise to 25 mL of potassium iodide (1.05 M) solution. The ion(s) present in very small quantity in the solution is/are
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 32
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: In the photoelectric effect, the electrons are ejected from the metal surface as soon as the beam of light of frequency greater than threshold frequency strikes the surface.
Reason R: When the photon of any energy strikes an electron in the atom, transfer of energy from the photon to the electron takes place.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 33
For compound having the formula GaAlCl$$_4$$, the correct option from the following is
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 34
For elements B, C, N, Li, Be, O and F, the correct order of first ionisation enthalpy is
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 35
Match List-I with List-II:
| List-I Species | List-II Geometry/Shape | ||
|---|---|---|---|
| A. | H$$_3$$O$$^+$$ | I. | Tetrahedral |
| B. | Acetylide anion | II. | Linear |
| C. | NH$$_4^+$$ | III. | Pyramidal |
| D. | ClO$$_2^-$$ | IV. | Bent |
Choose the correct answer from the options given below:
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 36
Match List-I with List-II:
| List-I | List-II | ||
|---|---|---|---|
| A. | K | I. | Thermonuclear reactions |
| B. | KCl | II. | Fertilizer |
| C. | KOH | III. | Sodium potassium pump |
| D. | Li | IV. | Absorbent of CO$$_2$$ |
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 37
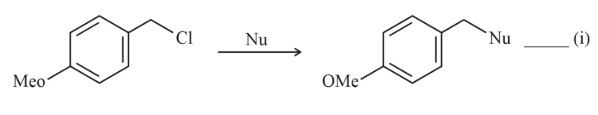
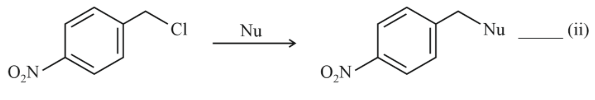
Where Nu = Nucleophile
Find out the correct statement from the options given below for the above two reactions.
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 38
Thin layer chromatography of a mixture shows the following observation.
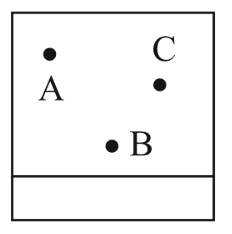
The correct order of elution in the silica gel column chromatography is
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 39
Arrange the following compounds in increasing order of rate of aromatic electrophilic substitution reaction.
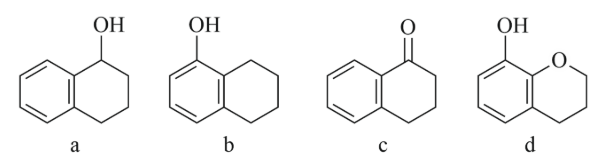
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 40
Given below are two statements:
Statement-I: If BOD is 4 ppm and dissolved oxygen is 8 ppm, then it is a good quality water.
Statement-II: If the concentration of zinc and nitrate salts are 5 ppm each, then it can be a good quality water.
In the light of the above statements, choose the most appropriate answer from the options below:
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 41
In the extraction process of copper, the product obtained after carrying out the reactions
(i) $$2Cu_2S + 3O_2 \to 2Cu_2O + 2SO_2$$
(ii) $$2Cu_2O + Cu_2S \to 6Cu + SO_2$$ is called
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 42
Given below are two statements:
Statement-I: Methane and steam passed over a heated Ni catalyst produces hydrogen gas.
Statement-II: Sodium nitrite reacts with NH$$_4$$Cl to give H$$_2$$O, N$$_2$$ and NaCl.
In the light of the above statements, choose the most appropriate answer from the options below:
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 43
When a solution of mixture of two inorganic salts was treated with freshly prepared ferrous sulphate in acidic medium, a dark brown ring was formed whereas on treatment with neutral FeCl$$_3$$, it gave deep red colour which disappeared on boiling and a brown red ppt was formed. The mixture contains
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 44
The complex that dissolves in water is
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 45
The set which does not have ambidentate ligand(s) is
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 46
Which of the following complex has a possibility to exist as meridional isomer?
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 47
L-isomer of tetrose X (C$$_4$$H$$_8$$O$$_4$$) gives positive Schiff's test and has two chiral carbons. On acetylation 'X' yields triacetate. 'X' also undergoes following reactions
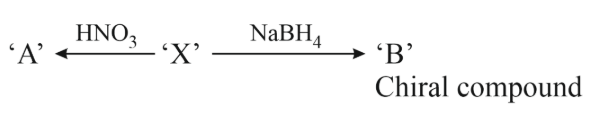
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 48
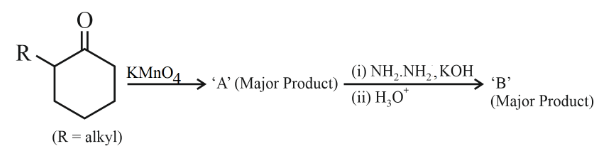
'A' and 'B' in the above reaction are :
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 49
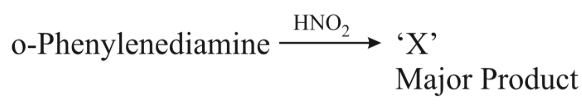
. 'X' is:
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 50
The polymer X-consists of linear molecules and is closely packed. It is prepared in the presence of trimethylaluminiumpropyl and titanium tetrachloride under low pressure. The polymer X is
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 51
A solution of sugar is obtained by mixing 200 g of its 25% solution and 500 g of its 40% solution (both by mass). The mass percentage of the resulting sugar solution is _______ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 52
Solid fuel used in rocket is a mixture of Fe$$_2$$O$$_3$$ and Al (in ratio 1 : 2). The heat evolved (kJ) per gram of the mixture is _______
Given $$\Delta H_f^0$$ Al$$_2$$O$$_3$$ = -1700 kJ mol$$^{-1}$$
$$\Delta H_f^0$$ Fe$$_2$$O$$_3$$ = -840 kJ mol$$^{-1}$$
Molar mass of Fe, Al and O are 56, 27 and 16 g mol$$^{-1}$$ respectively
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 53
A mixture of one mole of H$$_2$$O and 1 mole of CO is taken in a 10 litre container and heated to 725 K. At equilibrium 40% of water by mass reacts with carbon monoxide according to the equation:
$$CO(g) + H_2O(g) \rightleftharpoons CO_2(g) + H_2(g)$$
The equilibrium constant $$K_C \times 10^2$$ for the reaction is _______ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 54
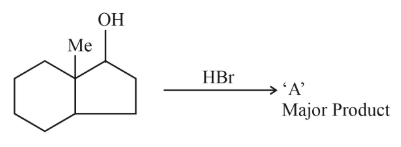
The number of hyperconjugation structures involved to stabilize carbocation formed in the reaction of 2-methylcyclohexan-1-ol with HBr is _______
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 55
An atomic substance A of molar mass 12 g mol$$^{-1}$$ has a cubic crystal structure with edge length of 300 pm. The no. of atoms present in one unit cell of A is _______ (Nearest integer)
Given the density of A is 3.0 g m m$$^{-1}$$ and NA$$_A$$ = $$6.02 \times 10^{23}$$ mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 56
0.004 M K$$_2$$SO$$_4$$ solution is isotonic with 0.01 M glucose solution. Percentage dissociation of K$$_2$$SO$$_4$$ is _______ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 57
In an electrochemical reaction of lead, at standard temperature, if $$E^0_{Pb^{2+}/Pb} = m$$ Volt and $$E^0_{Pb^{4+}/Pb} = n$$ Volt, then the value of $$E^0Pb^{2+}/Pb^{4+}$$ is given by $$m - xn$$. The value of x is _______ (Nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 58
KClO$$_3$$ + 6FeSO$$_4$$ + 3H$$_2$$SO$$_4$$ $$\to$$ KCl + 3Fe$$_2$$(SO$$_4$$)$$_3$$ + 3H$$_2$$O
The above reaction was studied at 300 K by monitoring the concentration of FeSO$$_4$$ in which initial concentration was 10 M and after half an hour became 8.8 M. The rate of production of Fe$$_2$$(SO$$_4$$)$$_3$$ is _______ $$\times 10^{-6}$$ mol L$$^{-1}$$ s$$^{-1}$$ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 59
The ratio of spin-only magnetic moment values $$\mu_{eff}[Cr(CN)_6]^{3-}$$ / $$\mu_{eff}[Cr(H_2O)_6]^{3+}$$ is _______
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Chemistry - Question 60
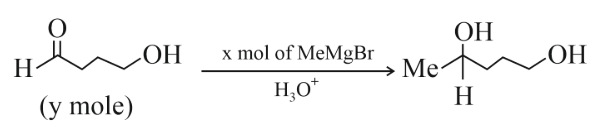
The ratio x/y on completion of the reaction is
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




