NTA JEE Main 11th April 2015 Online
For the following questions answer them individually
NTA JEE Main 11th April 2015 Online - Question 31
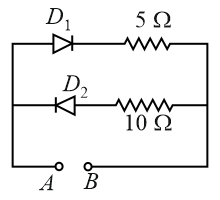
A 2 V battery is connected across AB as shown in the figure. The value of the current supplied by the battery when in first case battery's positive terminal is connected to A and in second case when positive terminal of battery is connected to B will respectively be:
NTA JEE Main 11th April 2015 Online - Question 32
In the following reaction:
$$A + 2B + 3C \rightleftharpoons AB_2C_3$$
6.0 g of A, $$6.0 \times 10^{23}$$ atoms of B and 0.036 mol of C reacted and formed 4.8 g of compound $$AB_2C_3$$. If the atomic mass of A and C are 60 and 80 amu, respectively. What is the atomic mass of B in amu? (Avogadro number = $$6 \times 10^{23}$$)
NTA JEE Main 11th April 2015 Online - Question 33
An element X shows +3, oxidation state in its compounds. Out of the four compounds given below, choose the incorrect formula for the element X.
NTA JEE Main 11th April 2015 Online - Question 34
At temperature T, the average kinetic energy of any particle is $$\frac{3}{2}kT$$. The de Broglie wavelength follows the order:
NTA JEE Main 11th April 2015 Online - Question 35
Molecule AB has a bond length of 1.617 $$\mathring{A}$$ and a dipole moment of 0.38 D. The fractional charge on each atom (absolute magnitude) is: $$(e_0 = 4.802 \times 10^{-10}$$ esu$$)$$
NTA JEE Main 11th April 2015 Online - Question 36
Which compound exhibits maximum dipole moment among the following?
NTA JEE Main 11th April 2015 Online - Question 37
When does a gas deviate the most from its ideal behavior?
NTA JEE Main 11th April 2015 Online - Question 38
The increase of pressure on ice $$\rightleftharpoons$$ water system at constant temperature will lead to:
NTA JEE Main 11th April 2015 Online - Question 39
Which physical property of di-hydrogen is wrong?
NTA JEE Main 11th April 2015 Online - Question 40
Which of the alkaline earth metal halides given below is essentially covalent in nature?


