NTA JEE Main 11th April 2014 Online
For the following questions answer them individually
NTA JEE Main 11th April 2014 Online - Question 41
Assuming that the degree of hydrolysis is small, the pH of 0.1M solution of sodium acetate (K$$_a$$ = $$1.0 \times 10^{-5}$$) will be:
NTA JEE Main 11th April 2014 Online - Question 42
Which of the following statements about Na$$_2$$O$$_2$$ is not correct?
NTA JEE Main 11th April 2014 Online - Question 43
The reagent needed for converting:
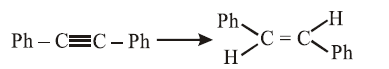
NTA JEE Main 11th April 2014 Online - Question 44
The gas liberated by the electrolysis of Dipotassium succinate solution is:
NTA JEE Main 11th April 2014 Online - Question 45
Which of the following statements about the depletion of ozone layer is correct?
NTA JEE Main 11th April 2014 Online - Question 46
The appearance of colour in solid alkali metal halides is generally due to:
NTA JEE Main 11th April 2014 Online - Question 47
In some solutions, the concentration of H$$_3$$O$$^+$$ remains constant even when small amounts of strong acid or strong base are added to them. These solutions are known as:
NTA JEE Main 11th April 2014 Online - Question 48
Given:
Fe$$^{3+}$$(aq) + e$$^-$$ → Fe$$^{2+}$$(aq); E$$^0$$ = +0.77 V
Al$$^{3+}$$(aq) + 3e$$^-$$ → Al(s); E$$^0$$ = $$-1.66$$ V
Br$$_2$$(aq) + 2e$$^-$$ → 2Br$$^-$$; E$$^0$$ = +1.09 V
Considering the electrode potentials, which of the following represents the correct order of reducing power?
NTA JEE Main 11th April 2014 Online - Question 49
In the reaction of formation of sulphur trioxide by contact process $$2SO_2 + O_2 \rightleftharpoons 2SO_3$$ the rate of reaction was measured as $$\frac{d[O_2]}{dt} = -2.5 \times 10^{-4}$$ mol L$$^{-1}$$ s$$^{-1}$$. The rate of reaction in terms of [SO$$_2$$] in mol L$$^{-1}$$ s$$^{-1}$$ will be:
NTA JEE Main 11th April 2014 Online - Question 50
For the reaction, $$2N_2O_5 \to 4NO_2 + O_2$$, the rate equation can be expressed in two ways:
$$-\frac{d[N_2O_5]}{dt} = k[N_2O_5]$$ and $$+\frac{d[NO_2]}{dt} = k'[N_2O_5]$$. k and k' are related as:


