NTA JEE Main 10th January 2019 Shift 2
For the following questions answer them individually
NTA JEE Main 10th January 2019 Shift 2 - Question 51
A reaction of cobalt (III) chloride and ethylenediamine in a 1 : 2 mole ratio generates two isomeric products A (violet-coloured) and B (green-coloured). A can show optical activity, but, B is optically inactive. What type of isomers do A and B represent?
NTA JEE Main 10th January 2019 Shift 2 - Question 52
The difference in the number of unpaired electrons of a metal ion in its high-spin and low-spin octahedral complexes is two. The metal ion is:
NTA JEE Main 10th January 2019 Shift 2 - Question 53
The major product of the following reaction is:
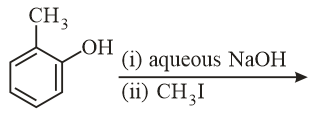
NTA JEE Main 10th January 2019 Shift 2 - Question 54
The major product obtained in the following reaction is:
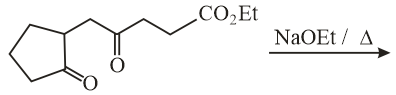
NTA JEE Main 10th January 2019 Shift 2 - Question 55
The major product of the following reaction is:
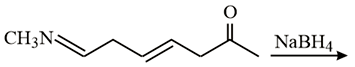
NTA JEE Main 10th January 2019 Shift 2 - Question 56
Which is the most suitable reagent for the following transformation?
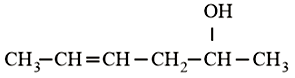
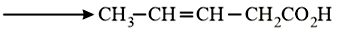
NTA JEE Main 10th January 2019 Shift 2 - Question 57
An aromatic compound 'A' having molecular formula $$C_7H_6O_2$$, on treating with aqueous ammonia and heating forms compound 'B'. The compound 'B' on reaction with molecular bromine and potassium hydroxide provides compound 'C' having molecular formula $$C_6H_7N$$. The structure of 'A' is:
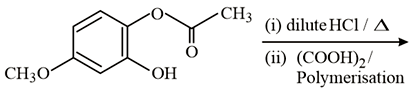
NTA JEE Main 10th January 2019 Shift 2 - Question 58
The major product of the following reaction is:
NTA JEE Main 10th January 2019 Shift 2 - Question 59
Which of the following tests cannot be used for identifying amino acids?
NTA JEE Main 10th January 2019 Shift 2 - Question 60
The correct match between item I and item II is:
Item I (Compound) Item II (Reagent)
a. Lysine p. 1-naphthol
b. Furfural q. Ninhydrin
c. Benzyl alcohol r. KMnO$$_4$$
d. Styrene s. Ceric ammonium nitrate


