NTA JEE Main 10th January 2019 Shift 2
For the following questions answer them individually
NTA JEE Main 10th January 2019 Shift 2 - Question 31
The 71$$^{st}$$ electron of an element X with an atomic number of 71 enters the orbital:
NTA JEE Main 10th January 2019 Shift 2 - Question 32
The ground state energy of a hydrogen atom is $$-13.6$$ eV. The energy of second excited state of He$$^+$$ ion in eV is:
NTA JEE Main 10th January 2019 Shift 2 - Question 33
The process with negative entropy change is:
NTA JEE Main 10th January 2019 Shift 2 - Question 34
An ideal gas undergoes isothermal compression from 5 m$$^3$$ to 1 m$$^3$$ against a constant external pressure of 4 N m$$^{-2}$$. The heat released in this process is 24 J mol$$^{-1}$$ K$$^{-1}$$ and is used to increase the pressure of 1 mole of Al. The temperature of Al increases by:
NTA JEE Main 10th January 2019 Shift 2 - Question 35
5.1 g $$NH_4SH$$ is introduced in 3.0 L evacuated flask at 327$$^{\circ}$$C. 30% of the solid $$NH_4SH$$ is decomposed to $$NH_3$$ and $$H_2S$$ as gases. The $$K_P$$ of the reaction at 327$$^{\circ}$$C is: ($$R = 0.082$$ L atm mol$$^{-1}$$ K$$^{-1}$$, Molar mass of S = 32 g mol$$^{-1}$$, Molar mass of N = 14 g mol$$^{-1}$$)
NTA JEE Main 10th January 2019 Shift 2 - Question 36
In the reaction of oxalate with permanganate in acidic medium, the number of electrons involved in producing one molecule of CO$$_2$$ is:
NTA JEE Main 10th January 2019 Shift 2 - Question 37
The number of 2-centre-2-electron and 3-centre-2-electron bonds in $$B_2H_6$$, respectively, are:
NTA JEE Main 10th January 2019 Shift 2 - Question 38
What is the IUPAC name of the following compound?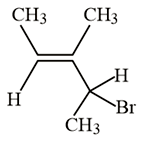
NTA JEE Main 10th January 2019 Shift 2 - Question 39
What will be the major product in the following mononitration reaction?
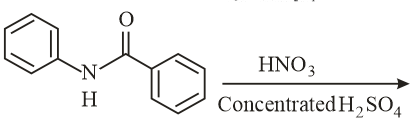
NTA JEE Main 10th January 2019 Shift 2 - Question 40
The reaction that is not involved in the ozone layer depletion mechanism in the stratosphere is:


