NTA JEE Main 10th January 2019 Shift 1
For the following questions answer them individually
NTA JEE Main 10th January 2019 Shift 1 - Question 41
The increasing order of the pKa values of the following compounds is:
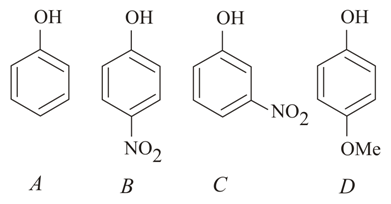
NTA JEE Main 10th January 2019 Shift 1 - Question 42
If Dichloromethane (DCM) and water $$H_2O$$ are used for differential extraction, which one of the following statements is correct?
NTA JEE Main 10th January 2019 Shift 1 - Question 43
The major product of the following reaction is:
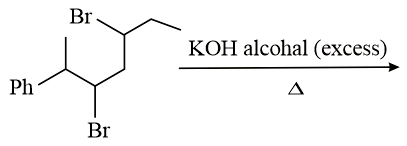
NTA JEE Main 10th January 2019 Shift 1 - Question 44
Which hydrogen in compound (E) is easily replaceable during bromination reaction in presence of light?
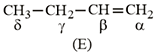
NTA JEE Main 10th January 2019 Shift 1 - Question 45
Water filled in two glasses A and B gave BOD values of 10 and 20, respectively. The correct statement regarding them is:
NTA JEE Main 10th January 2019 Shift 1 - Question 46
Which primitive unit cell has unequal edge lengths ($$a \neq b \neq c$$) and all axial angles different from 90$$^{\circ}$$?
NTA JEE Main 10th January 2019 Shift 1 - Question 47
Liquids A and B form an ideal solution in the entire composition range. At 350K, the vapour pressure of pure A and pure B are $$7 \times 10^3$$ Pa and $$12 \times 10^3$$ Pa, respectively. The composition of the vapour in equilibrium with a solution containing 40 mole percent of A at this temperature is:
NTA JEE Main 10th January 2019 Shift 1 - Question 48
Consider the following reduction processes:
$$Zn^{2+} + 2e^- \rightarrow Zn(s); E^{\circ} = -0.76$$ V
$$Ca^{2+} + 2e^- \rightarrow Ca(s); E^{\circ} = -2.87$$ V
$$Mg^{2+} + 2e^- \rightarrow Mg(s); E^{\circ} = -2.36$$ V
$$Ni^{2+} + 2e^- \rightarrow Ni(s); E^{\circ} = -0.25$$ V
The reducing power of the metals increases in the order:
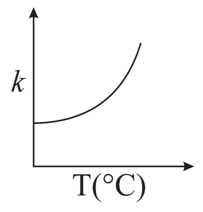
NTA JEE Main 10th January 2019 Shift 1 - Question 49
Consider the given plots for a reaction obeying Arrhenius equation ($$0^{\circ}$$C < T < 300$$^{\circ}$$C): (K and $$E_a$$ are rate constant and activation energy, respectively)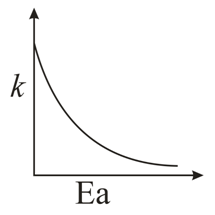
NTA JEE Main 10th January 2019 Shift 1 - Question 50
Which of the following is not an example of heterogeneous catalysis reaction?


