NTA JEE Main 10th April 2023 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 31
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: 3.1500 g of hydrated oxalic acid dissolved in water to make 250.0 mL solution will result in 0.1 M oxalic acid solution.
Reason R: Molar mass of hydrated oxalic acid is 126 g mol$$^{-1}$$.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 32
Match List-I with List-II
| List-I | List-II | ||
|---|---|---|---|
| A | 16 g of CH$$_4$$(g) | I | Weighs 28g |
| B | 1 g of H$$_2$$(g) | II | $$60.2 \times 10^{23}$$ electrons |
| C | 1 mole of N$$_2$$(g) | III | Weighs 32g |
| D | 0.5 mol of SO$$_2$$(g) | IV | Occupies 11.4 L volume at STP |
Choose the correct answer from the options given below:
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 33
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: The energy required to form Mg$$^{2+}$$ from Mg is much higher than that required to produce Mg$$^+$$
Reason R: Mg$$^{2+}$$ is small ion and carry more charge than Mg$$^+$$
In the light of the above statements, choose the correct answer from the options given below.
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 34
The correct order of metallic character is
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 35
Gibbs energy vs T plot for the formation of oxides is given below. For the given diagram, the correct statement is-
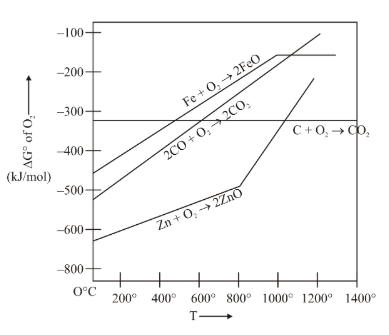
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 36
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Physical properties of isotopes of hydrogen are different.
Reason R: Mass difference between isotopes of hydrogen is very large.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 37
Number of water molecules in washing soda and soda ash respectively are:
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 38
The decreasing order of hydride affinity for following carbocations is:
(a)
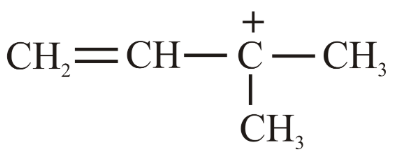
(b)
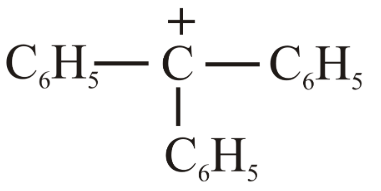
(c)
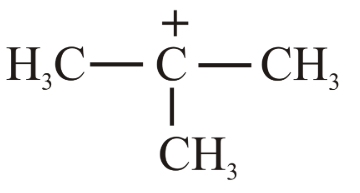
(d)
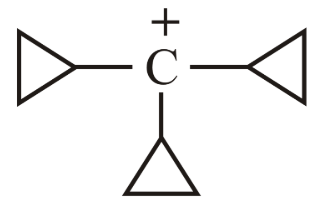
Choose the correct answer from the options given below:
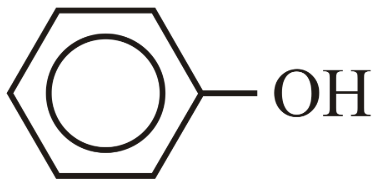
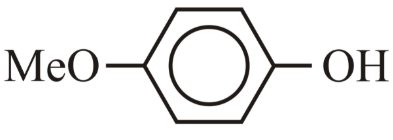
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 39
The correct order for acidity of the following hydroxyl compound is
(A) CH$$_3$$OH
(B) (CH$$_3$$)$$_3$$COH
(C)
(D)
(E)
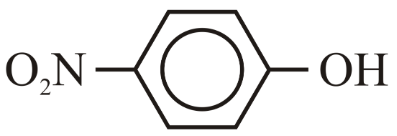
Choose the correct answer from the options given below:
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 40
In Carius tube, an organic compound 'X' is treated with sodium peroxide to form a mineral acid 'Y'. The solution of BaCl$$_2$$ is added to 'Y' to form a precipitate 'Z'. 'Z' is used for the quantitative estimation of an extra element. 'X' could be
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 41
The delicate balance of CO$$_2$$ and O$$_2$$ is NOT disturbed by
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 42
The correct relationships between unit cell edge length 'a' and radius of sphere 'r' for face-centred and body-centred cubic structures respectively are:
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 43
Ferric chloride is applied to stop bleeding because
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 44
Match List-I with List-II.
| List-I Complex | List-II Crystal Field splitting energy ($$\Delta_o$$) | ||
|---|---|---|---|
| A. | $$[Ti(H_2O)_6]^{2+}$$ | I. | -1.2 |
| B. | $$[V(H_2O)_6]^{2+}$$ | II. | -0.6 |
| C. | $$[Mn(H_2O)_6]^{3+}$$ | III. | 0 |
| D. | $$[Fe(H_2O)_6]^{3+}$$ | IV. | -0.8 |
Choose the correct answer from the options given below:
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 45
The correct order of the number of unpaired electrons in the given complexes is
(A) $$[Fe(CN)_6]^{3-}$$
(B) $$FeF_6^{3-}$$
(C) $$CoF_6^{3-}$$
(D) $$[Cr(oxalate)_3]^{3-}$$
(E) $$Ni(CO)_4$$
Choose the correct answer from the options given below:
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 46
The major product 'P' formed in the given reaction is
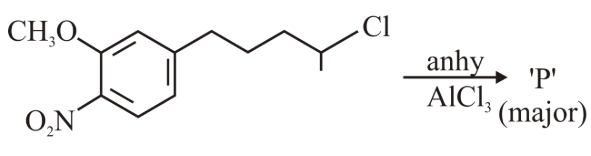
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 47
Incorrect method of preparation for alcohols from the following is:
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 48
In the reaction given below, the product 'X' is:
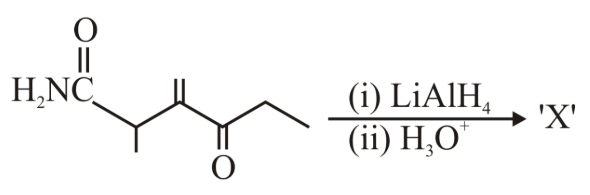
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 49
Buna-S can be represented as:
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 50
The reaction used for preparation of soap from fat is:
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 51
The electron in the n$$^{th}$$ orbit of Li$$^{2+}$$ is excited to (n+1) orbit using the radiation of energy $$1.47 \times 10^{-17}$$ J (as shown in the diagram). The value of n is _______ Given: $$R_H = 2.18 \times 10^{-18}$$ J
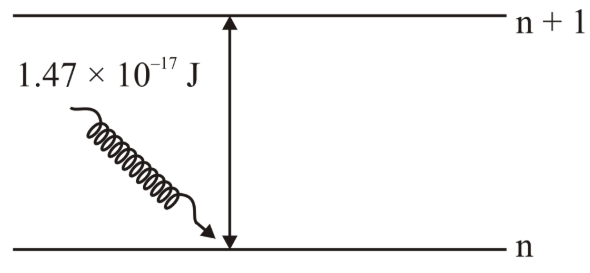
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 52
For a metal ion, the calculated magnetic moment is 4.90 BM. This metal ion has _______ number of unpaired electrons
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 53
The number of molecules from the following which contain only two lone pair of electrons is _______
H$$_2$$O, N$$_2$$, CO, XeF$$_4$$, NH$$_3$$, NO, CO$$_2$$, F$$_2$$
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 54
$$A(g) \rightleftharpoons 2B(g) + C(g)$$
For the given reaction, if the initial pressure is 450 mmHg and the pressure at time t is 720 mmHg at a constant temperature T and constant volume V. The fraction of A(g) decomposed under these conditions is $$x \times 10^{-1}$$. The value of x is (nearest integer) _______
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 55
The number of endothermic process/es from the following is _______.
A. $$I_2(g) \to 2I(g)$$
B. $$HCl(g) \to H(g) + Cl(g)$$
C. $$H_2O(l) \to H_2O(g)$$
D. $$C(s) + O_2(g) \to CO_2(g)$$
E. Dissolution of ammonium chloride in water
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 56
In alkaline medium, the reduction of permanganate anion involves a gain of _______ electrons.
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 57
An aqueous solution of volume 300 cm$$^3$$ contains 0.63 g of protein. The osmotic pressure of the solution at 300 K is 1.29 mbar. The molar mass of the protein is _______ g mol$$^{-1}$$.
Given: R = 0.083 L bar K$$^{-1}$$ mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 58
The specific conductance of 0.0025M acetic acid is $$5 \times 10^{-5}$$ S cm$$^{-1}$$ at a certain temperature. The dissociation constant of acetic acid is _______ $$\times 10^{-7}$$. (Nearest integer)
Consider limiting molar conductivity of CH$$_3$$COOH as 400 S cm$$^2$$ mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 59
The number of incorrect statement/s from the following is _______
A. The successive half lives of zero order reactions decreases with time.
B. A substance appearing as reactant in the chemical equation may not affect the rate of reaction
C. Order and molecularity of a chemical reaction can be a fractional number
D. The rate constant units of zero and second order reaction are mol L$$^{-1}$$ s$$^{-1}$$ and mol$$^{-1}$$ L s$$^{-1}$$ respectively
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 2 - Chemistry - Question 60
The difference in the oxidation state of Xe between the oxidised product of Xe formed on complete hydrolysis of XeF$$_4$$ and XeF$$_4$$ is _______
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




