NTA JEE Main 10th April 2023 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 31
The number of molecules and moles in 2.8375 litres of O$$_2$$ at STP are respectively
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 32
The pair from the following pairs having both compounds with net non-zero dipole moment is
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 33
The compound which does not exist is
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 34
The enthalpy change for the adsorption process and micelle formation respectively are
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 35
Given
(A) $$2CO(g) + O_2(g) \to 2CO_2(g)$$, $$\Delta H_1^0 = -x$$ kJ mol$$^{-1}$$
(B) $$C_{graphite} + O_2(g) \to CO_2(g)$$, $$\Delta H_2^0 = -y$$ kJ mol$$^{-1}$$
The $$\Delta H^0$$ for the reaction $$C_{graphite} + \frac{1}{2}O_2(g) \to CO(g)$$ is
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 36
Given below are two reactions, involved in the commercial production of dihydrogen H$$_2$$. The two reactions are carried out at temperature "T$$_1$$" and "T$$_2$$", respectively
$$C(s) + H_2O(g) \xrightarrow{T_1} CO(g) + H_2(g)$$
$$CO(g) + H_2O(g) \xrightarrow{T_2, Catalyst} CO_2(g) + H_2(g)$$
The temperatures T$$_1$$ and T$$_2$$ are correctly related as
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 37
Lime reacts exothermally with water to give 'A' which has low solubility in water. Aqueous solution of 'A' is often used for the test of CO$$_2$$, a test in which insoluble B is formed. If B is further reacted with CO$$_2$$ then soluble compound is formed. 'A' is
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 38
Using column chromatography, mixture of two compounds 'A' and 'B' was separated. 'A' eluted first, this indicates 'B' has
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 39
The major product 'P' formed in the given reaction is
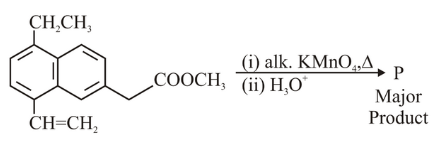
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 40
Match List I with List II
| List I Industry | List II Waste Generated | ||
|---|---|---|---|
| (A) | Steel plants | (I) | Gypsum |
| (B) | Thermal power plants | (II) | Fly ash |
| (C) | Fertilizer Industries | (III) | Slag |
| (D) | Paper mills | (IV) | Bio-degradable wastes |
Choose the correct answer from the options below:
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 41
Which of the following is used as a stabilizer during the concentration of sulphide ores?
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 42
Given below are two statements:
Statement I: Aqueous solution of K$$_2$$Cr$$_2$$O$$_7$$ is preferred as a primary standard in volumetric analysis over Na$$_2$$Cr$$_2$$O$$_7$$ aqueous solution.
Statement II: K$$_2$$Cr$$_2$$O$$_7$$ has a higher solubility in water than Na$$_2$$Cr$$_2$$O$$_7$$. In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 43
Prolonged heating is avoided during the preparation of ferrous ammonium sulphate to
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 44
Which of the following statements are correct?
(A) The M$$^{3+}$$/M$$^{2+}$$ reduction potential for iron is greater than manganese.
(B) The higher oxidation states of first row d-block elements get stabilized by oxide ion
(C) Aqueous solution of Cr$$^{2+}$$ can liberate hydrogen from dilute acid
(D) Magnetic moment of V$$^{2+}$$ is observed between 4.4-5.2 BM
Choose the correct answer from the options given below:
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 45
The octahedral diamagnetic low spin complex among the following is
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 46
Identify the correct order of reactivity for the following pairs towards the respective mechanism
(A)
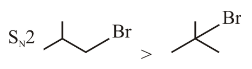
(B)
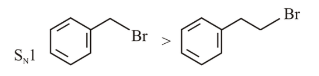
(C) Electrophilic substitution
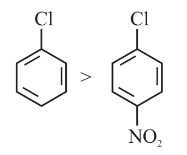
(D) Nucleophilic substitution
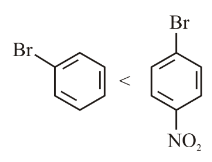
Choose the correct answer from the options given below:
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 47
Suitable reaction condition for preparation of Methyl phenyl ether is
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 48
Isomeric amines with molecular formula C$$_8$$H$$_{11}$$N give the following tests
Isomer P $$\Rightarrow$$ Can be prepared by Gabriel phthalimide synthesis
Isomer Q $$\Rightarrow$$ Reacts with Hinsberg's reagent to give solid insoluble in NaOH
Isomer R $$\Rightarrow$$ Reacts with HONO followed by $$\beta$$-naphthol in NaOH to give red dye.
Isomers P, Q and R respectively are
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 49
Match List-I with List-II.
| List-I Polymer | List-II Type/Class | ||
|---|---|---|---|
| A. | Nylon-2-Nylon-6 | I. | Thermosetting polymer |
| B. | Buna-N | II. | Biodegradable polymer |
| C. | Ureaformaldehyde resin | III. | Synthetic rubber |
| D. | Dacron | IV. | Polyester |
Choose the correct answer from the options given below:
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 50
The one that does not stabilize 2$$^\circ$$ and 3$$^\circ$$ structures of proteins is
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 51
The number of bent-shaped molecule/s from the following is _______ N$$_3^-$$, NO$$_2$$, I$$_3^-$$, O$$_3$$, SO$$_2$$
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 52
The sum of lone pairs present on the central atom of the interhalogen IF$$_5$$ and IF$$_7$$ is _______.
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 53
At constant temperature, a gas is at a pressure of 940.3 mm Hg. The pressure at which its volume decreases by 40% is _______ mm Hg. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 54
$$FeO_4^{2-} \xrightarrow{+2.2 V} Fe^{3+} \xrightarrow{+0.70 V} Fe^{2+} \xrightarrow{-0.45 V} Fe^0$$
$$E^0_{FeO_4^{2-}/Fe^{2+}}$$ is $$x \times 10^{-3}$$ V. The value of x is _______.
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 55
The number of incorrect statement/s about the black body from the following is _______.
(A) Emit or absorb energy in the form of electromagnetic radiation.
(B) Frequency distribution of the emitted radiation depends on temperature.
(C) At a given temperature, intensity vs frequency curve passes through a maximum value.
(D) The maximum of the intensity vs frequency curve is at a higher frequency at higher temperature compared to that at lower temperature.
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 56
The number of correct statement/s involving equilibria in physical processes from the following is _______.
(A) Equilibrium is possible only in a closed system at a given temperature.
(B) Both the opposing processes occur at the same rate.
(C) When equilibrium is attained at a given temperature, the value of all its parameters became equal
(D) For dissolution of solids in liquids, the solubility is constant at a given temperature.
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 57
In the following reaction, the total number of oxygen atoms in X and Y is _______.
Na$$_2$$O + H$$_2$$O $$\to$$ 2X
Cl$$_2$$O$$_7$$ + H$$_2$$O $$\to$$ 2Y
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 58
If the degree of dissociation of aqueous solution of weak monobasic acid is determined to be 0.3, then the observed freezing point will be _______ % higher than the expected/theoretical freezing point. (Nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 59
A molecule undergoes two independent first order reactions whose respective half lives are 12 min and 3 min. If both the reactions are occurring then the time taken for the 50% consumption of the reactant is _______ min. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 10th April 2023 Shift 1 - Chemistry - Question 60
In potassium ferrocyanide, there are _______ pairs of electrons in the t$$_{2g}$$ set of orbitals.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




