NTA JEE Main 10th April 2019 Shift 1
For the following questions answer them individually
NTA JEE Main 10th April 2019 Shift 1 - Question 31
At 300 K and 1 atmospheric pressure, 10 mL of a hydrocarbon required 55 mL of O$$_2$$ for complete combustion, and 40 mL of CO$$_2$$ is formed. The formula of the hydrocarbon is:
NTA JEE Main 10th April 2019 Shift 1 - Question 32
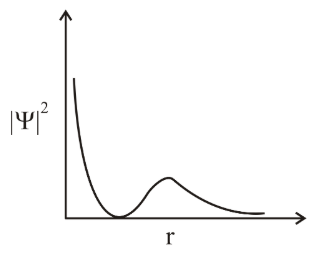
The graph between $$|\psi|^2$$ and r (radical distance) is shown below. This represents:
NTA JEE Main 10th April 2019 Shift 1 - Question 33
The isoelectronic set of ions is:
NTA JEE Main 10th April 2019 Shift 1 - Question 34
During the change of O$$_2$$ to O$$_2^-$$, the incoming electron goes to the orbital:
NTA JEE Main 10th April 2019 Shift 1 - Question 35
Consider the following table:
Gas a/(kPa dm$$^6$$ mol$$^{-1}$$) b/(dm$$^3$$ mol$$^{-1}$$)
A 642.32 0.05196
B 155.21 0.04136
C 431.91 0.05196
D 155.21 0.4382
a and b are van der Waals constants. The correct statement about the gases is:
NTA JEE Main 10th April 2019 Shift 1 - Question 36
A process will be spontaneous at all temperatures if:
NTA JEE Main 10th April 2019 Shift 1 - Question 37
Consider the following statements
(a) The pH of a mixture containing 400 mL of 0.1 M H$$_2$$SO$$_4$$ and 400 mL of 0.1 M NaOH will be approximately 1.3.
(b) Ionic product of water is temperature dependent.
(c) A monobasic acid with K$$_a$$ = 10$$^{-5}$$ has a pH = 5. The degree of dissociation of this acid is 50%.
(d) The Le Chatelier's principle is not applicable to common-ion effect.
The correct statements are:
NTA JEE Main 10th April 2019 Shift 1 - Question 38
The synonym for water gas when used in the production of methanol is:
NTA JEE Main 10th April 2019 Shift 1 - Question 39
The alloy used in the construction of aircrafts is:
NTA JEE Main 10th April 2019 Shift 1 - Question 40
The correct order of catenation is:


