NTA JEE Main 10th April 2016 Online
For the following questions answer them individually
NTA JEE Main 10th April 2016 Online - Question 41
Sodium extract is heated with concentrated $$HNO_3$$ before testing for halogens because:
NTA JEE Main 10th April 2016 Online - Question 42
Consider the reaction sequence below:
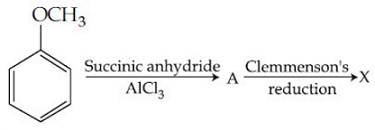
X is:
NTA JEE Main 10th April 2016 Online - Question 43
Which one of the following substances used in dry cleaning is a better strategy to control environmental pollution?
NTA JEE Main 10th April 2016 Online - Question 44
An aqueous solution of a salt $$MX_2$$ at certain temperature has a Van't Hoff factor of 2. What is the degree of dissociation for this solution of the salt?
NTA JEE Main 10th April 2016 Online - Question 45
Oxidation of succinate ion produces ethylene and carbon dioxide gases. On passing 0.2 Faraday electricity through an aqueous solution of potassium succinate, what is the total volume of gases (at both cathode and anode) at STP (1 atm and 273 K)?
NTA JEE Main 10th April 2016 Online - Question 46
Identify the correct statement:
NTA JEE Main 10th April 2016 Online - Question 47
The rate law for the reaction below is given by the expression k[A][B]
$$A + B \to$$ Product
If the concentration of B is increased from 0.1 to 0.3 mol, keeping the value of A at 0.1 mol, the rate constant will be:
NTA JEE Main 10th April 2016 Online - Question 48
Gold numbers of some colloids are Gelatin: 0.005 - 0.01, Gum Arabic: 0.15 - 0.25, Oleate: 0.04 - 1.0, Starch: 15 - 25. Which among the following is a better protective colloid?
NTA JEE Main 10th April 2016 Online - Question 49
Extraction of copper by smelting uses silica as an additive to remove.
NTA JEE Main 10th April 2016 Online - Question 50
Identify the incorrect statement:


