NTA JEE Main 1st September 2021 Shift 2
For the following questions answer them individually
NTA JEE Main 1st September 2021 Shift 2 - Question 41
The oxide without nitrogen-nitrogen bond is:
NTA JEE Main 1st September 2021 Shift 2 - Question 42
In the given chemical reaction, colors of the Fe$$^{2+}$$ and Fe$$^{3+}$$ ions, are respectively:
$$5Fe^{2+} + MnO_4^- + 8H^+ \rightarrow Mn^{2+} + 4H_2O + 5Fe^{3+}$$
NTA JEE Main 1st September 2021 Shift 2 - Question 43
Identify the element for which electronic configuration in +3 oxidation state is [Ar]3d$$^5$$:
NTA JEE Main 1st September 2021 Shift 2 - Question 44
The Crystal Field Stabilization Energy (CFSE) and magnetic moment (spin-only) of an octahedral aqua complex of a metal ion M$$^{Z+}$$ are $$-0.8\Delta_0$$ and 3.87 BM, respectively. Identify M$$^{Z+}$$:
NTA JEE Main 1st September 2021 Shift 2 - Question 45
Experimentally reducing a functional group cannot be done by which one of the following reagents?
NTA JEE Main 1st September 2021 Shift 2 - Question 46
Given below are two statements:
Statement I : The nucleophilic addition of sodium hydrogen sulphite to an aldehyde or a ketone involves proton transfer to form a stable ion.
Statement II : The nucleophilic addition of hydrogen cyanide to an aldehyde or a ketone yields amine as final product.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 1st September 2021 Shift 2 - Question 47
In the following sequence of reactions a compound A, (molecular formula C$$_6$$H$$_{12}$$O$$_2$$) with a straight chain structure gives a C$$_4$$ carboxylic acid. A is:
$$A \xrightarrow[\mathrm{H_3O^+}]{\mathrm{LiAlH_4}} B \xrightarrow{\text{Oxidation}} \text{C}_4\text{-carboxylic acid}$$
NTA JEE Main 1st September 2021 Shift 2 - Question 48
Which one of the following gives the most stable Diazonium salt?
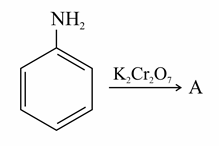
NTA JEE Main 1st September 2021 Shift 2 - Question 49
Identify A in the following reaction.
NTA JEE Main 1st September 2021 Shift 2 - Question 50
Monomer units of Dacron polymer are:


