NTA JEE Main 8th April 2023 Shift 2
For the following questions answer them individually
NTA JEE Main 8th April 2023 Shift 2 - Question 51
For complete combustion of ethene,
$$C_2H_4(g) + 3O_2(g) \to 2CO_2(g) + 2H_2O(l)$$
the amount of heat produced as measured in bomb calorimeter is 1406 kJ mol$$^{-1}$$ at 300 K. The minimum value of T$$\Delta$$S needed to reach equilibrium is ($$-$$) kJ. (Nearest integer)
Given: R = 8.3 J K$$^{-1}$$ mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 8th April 2023 Shift 2 - Question 52
The solubility product of BaSO$$_4$$ is $$1 \times 10^{-10}$$ at 298 K. The solubility of BaSO$$_4$$ in 0.1 M K$$_2$$SO$$_4$$(aq) solution is _______ $$\times 10^{-9}$$ g L$$^{-1}$$ (nearest integer).
Given: Molar mass of BaSO$$_4$$ is 233 g mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 8th April 2023 Shift 2 - Question 53
If the boiling points of two solvents X and Y (having same molecular weights) are in the ratio 2:1 and their enthalpy of vaporizations are in the ratio 1:2, then the boiling point elevation constant of X is m times the boiling point elevation constant of Y. The value of m is (nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 8th April 2023 Shift 2 - Question 54
Coagulating value of the electrolytes AlCl$$_3$$ and NaCl for As$$_2$$S$$_3$$ are 0.09 and 50.04 respectively. The coagulating power of AlCl$$_3$$ is x times the coagulating power of NaCl. The value of x is _____.
789
456
123
0.-
Clear All
NTA JEE Main 8th April 2023 Shift 2 - Question 55
The ratio of sigma and $$\pi$$ bonds present in pyrophosphoric acid is _____.
789
456
123
0.-
Clear All
NTA JEE Main 8th April 2023 Shift 2 - Question 56
The observed magnetic moment of the complex $$[Mn(NCS)_6]^{x-}$$ is 6.06 BM. The numerical value of x is _____.
789
456
123
0.-
Clear All
NTA JEE Main 8th April 2023 Shift 2 - Question 57
The sum of oxidation state of the metals in Fe(CO)$$_5$$, VO$$^{2+}$$ and WO$$_3$$ is _____.
789
456
123
0.-
Clear All
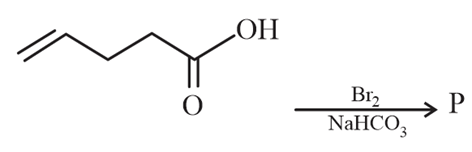
NTA JEE Main 8th April 2023 Shift 2 - Question 58
Major product 'P' formed in the following reaction is:
NTA JEE Main 8th April 2023 Shift 2 - Question 59
The product (P) formed from the following multistep reaction is:
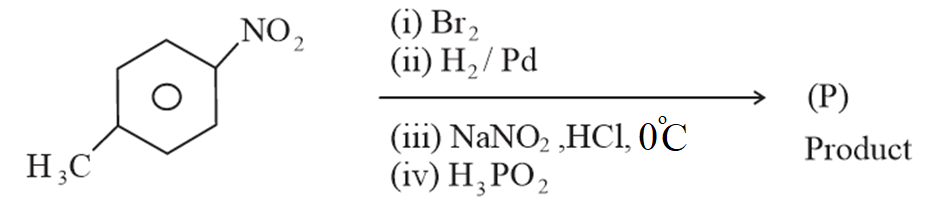
NTA JEE Main 8th April 2023 Shift 2 - Question 60
Match List I with List II
| List I Natural amino acid | List II One Letter Code | ||
|---|---|---|---|
| (A) | Glutamic acid | (I) | Q |
| (B) | Glutamine | (II) | W |
| (C) | Tyrosine | (III) | E |
| (D) | Tryptophan | (IV) | Y |
Choose the correct answer from the options given below:


