NTA JEE Main 8th April 2023 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 31
Which of the following have same number of significant figures?
(A) 0.00253
(B) 1.0003
(C) 15.0
(D) 163
Choose the correct answer from the options given below
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 32
Henry Moseley studied characteristic X-ray spectra of elements. The graph which represents his observation correctly is
Given $$\nu$$ = Frequency of X-ray emitted, $$Z$$ = Atomic number
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 33
The number of atomic orbitals from the following having 5 radial nodes is
7s, 7p, 6s, 8p, 8d
789
456
123
0.-
Clear All
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 34
The number of species from the following carrying a single lone pair on central atom Xenon is
$$XeF_5^+$$, $$XeO_3$$, $$XeO_2F_2$$, $$XeF_5^-$$, $$XeO_3F_2$$, $$XeOF_4$$, $$XeF_4$$
789
456
123
0.-
Clear All
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 35
Arrange the following gases in increasing order of van der Waals constant 'a'
(A) Ar
(B) CH$$_4$$
(C) H$$_2$$O
(D) C$$_6$$H$$_6$$
Choose the correct option from the following.
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 36
The incorrect statements from the following is:
A. The electrical work that a reaction can perform at constant pressure and temperature is equal to the reaction Gibbs energy.
B. $$E^\circ_{cell}$$ is dependent on the pressure.
C. $$\frac{dE_{cell}}{dT} = \frac{\Delta_r S}{nF}$$
D. A cell is operating reversibly if the cell potential is exactly balanced by an opposing source of potential difference.
789
456
123
0.-
Clear All
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 37
Given below are two statements:
Statement-I: Methyl orange is a weak acid.
Statement-II: The benzenoid form of methyl orange is more intense/deeply coloured than the quinonoid form.
In the light of the above statement, choose the most appropriate answer from the options given below:
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 38
Given below are two statements:
Statement I: In redox titration, the indicators used are sensitive to change in pH of the solution.
Statement II: In acid-base titration, the indicators used are sensitive to change in oxidation potential.
In the light of the above statements, choose the most appropriate answer from the options given below
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 39
Which of the following can reduce decomposition of H$$_2$$O$$_2$$ on exposure to light
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 40
For a good quality cement, the ratio of lime to the total of the oxides of Si, Al and Fe should be as close as to
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 41
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Sodium is about 30 times as abundant as potassium in the oceans.
Reason R: Potassium is bigger in size than sodium.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 42
The descending order of acidity for the following carboxylic acid is-
(A) CH$$_3$$COOH
(B) F$$_3$$C-COOH
(C) ClCH$$_2$$-COOH
(D) FCH$$_2$$-COOH
(E) BrCH$$_2$$-COOH
Choose the correct answer from the options given below:
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 43
The correct IUPAC nomenclature for the following compound is:
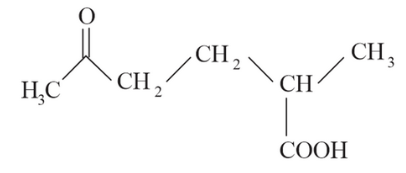
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 44
Which of these reactions is not a part of breakdown of ozone in stratosphere?
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 45
The correct reaction profile diagram for a positive catalyst reaction.
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 46
The statement/s which are true about antagonists from the following is/are:
A. They bind to the receptor site.
B. Get transferred inside the cell for their action.
C. Inhibit the natural communication of the body.
D. Mimic the natural messenger.
Choose the correct option from the options given below:
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 47
In Hall-Heroult process, the following is used for reducing Al$$_2$$O$$_3$$:-
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 48
Match List-I with List-II
| LIST-I Coordination Complex | LIST-II Number of unpaired electrons | ||
|---|---|---|---|
| A. | $$[Cr(CN)_6]^{3-}$$ | I. | 0 |
| B. | $$[Fe(H_2O)_6]^{2+}$$ | II. | 3 |
| C. | $$[Co(NH_3)_6]^{3+}$$ | III. | 2 |
| D. | $$[Ni(NH_3)_6]^{2+}$$ | IV. | 4 |
Choose the correct answer from the options given below:
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 49
The correct order of reactivity of following haloarenes towards nucleophilic substitution with aqueous NaOH is:
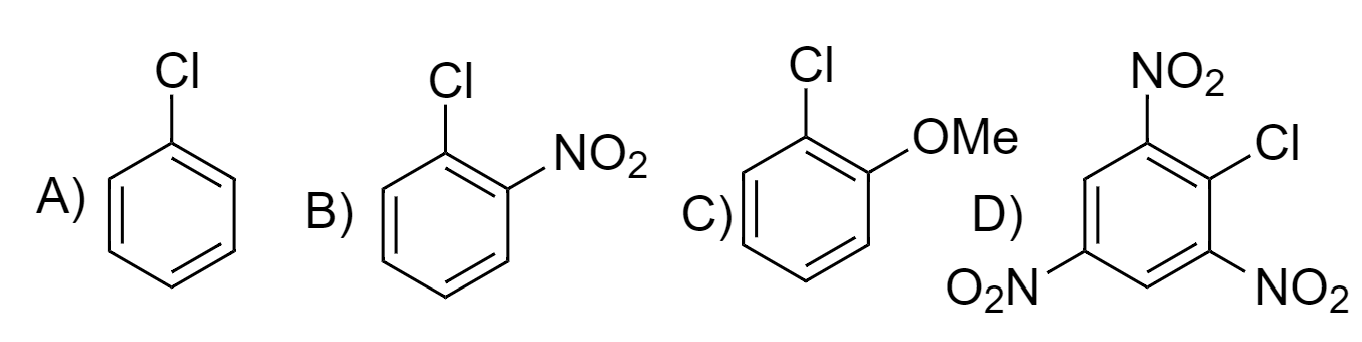
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 50
A compound 'X' when treated with phthalic anhydride in presence of concentrated H$$_2$$SO$$_4$$ yields 'Y'. 'Y' is used as an acid/base indicator. 'X' and 'Y' are respectively
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 51
For complete combustion of ethene,
$$C_2H_4(g) + 3O_2(g) \to 2CO_2(g) + 2H_2O(l)$$
the amount of heat produced as measured in bomb calorimeter is 1406 kJ mol$$^{-1}$$ at 300 K. The minimum value of T$$\Delta$$S needed to reach equilibrium is ($$-$$) kJ. (Nearest integer)
Given: R = 8.3 J K$$^{-1}$$ mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 52
The solubility product of BaSO$$_4$$ is $$1 \times 10^{-10}$$ at 298 K. The solubility of BaSO$$_4$$ in 0.1 M K$$_2$$SO$$_4$$(aq) solution is _______ $$\times 10^{-9}$$ g L$$^{-1}$$ (nearest integer).
Given: Molar mass of BaSO$$_4$$ is 233 g mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 53
If the boiling points of two solvents X and Y (having same molecular weights) are in the ratio 2:1 and their enthalpy of vaporizations are in the ratio 1:2, then the boiling point elevation constant of X is m times the boiling point elevation constant of Y. The value of m is (nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 54
Coagulating value of the electrolytes AlCl$$_3$$ and NaCl for As$$_2$$S$$_3$$ are 0.09 and 50.04 respectively. The coagulating power of AlCl$$_3$$ is x times the coagulating power of NaCl. The value of x is _____.
789
456
123
0.-
Clear All
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 55
The ratio of sigma and $$\pi$$ bonds present in pyrophosphoric acid is _____.
789
456
123
0.-
Clear All
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 56
The observed magnetic moment of the complex $$[Mn(NCS)_6]^{x-}$$ is 6.06 BM. The numerical value of x is _____.
789
456
123
0.-
Clear All
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 57
The sum of oxidation state of the metals in Fe(CO)$$_5$$, VO$$^{2+}$$ and WO$$_3$$ is _____.
789
456
123
0.-
Clear All
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 58
Major product 'P' formed in the following reaction is:
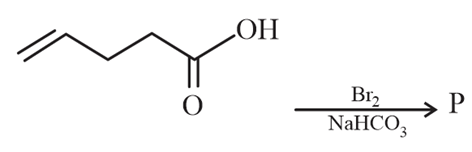
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 59
The product (P) formed from the following multistep reaction is:
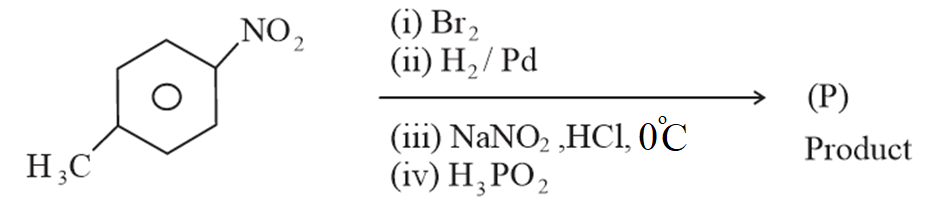
NTA JEE Main 8th April 2023 Shift 2 - Chemistry - Question 60
Match List I with List II
| List I Natural amino acid | List II One Letter Code | ||
|---|---|---|---|
| (A) | Glutamic acid | (I) | Q |
| (B) | Glutamine | (II) | W |
| (C) | Tyrosine | (III) | E |
| (D) | Tryptophan | (IV) | Y |
Choose the correct answer from the options given below:
.webp)
.webp)
.webp)
.webp)




