NTA JEE Main 1st February 2023 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 31
Which one of the following sets of ions represents a collection of isoelectronic species?
(Given: Atomic Number: F: 9, Cl: 17, Na = 11, Mg = 12, Al = 13, K = 19, Ca = 20, Sc = 21)
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 32
For electron gain enthalpies of the elements denoted as $$\Delta_{eg}$$H, the incorrect option is:
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 33
The effect of addition of helium gas to the following reaction in equilibrium state at constant volume, is:
$$\text{PCl}_5(g) \rightleftharpoons \text{PCl}_3(g) + \text{Cl}_2(g)$$
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 34
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A): An aqueous solution of KOH when for volumetric analysis, its concentration should be checked before the use.
Reason (R): On aging, KOH solution absorbs atmospheric CO$$_2$$.
In the light of the above statements, choose the correct answer from the options given below.
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 35
O $$-$$ O bond length in H$$_2$$O$$_2$$ is X than the O $$-$$ O bond length in F$$_2$$O$$_2$$. The O $$-$$ H bond length in H$$_2$$O$$_2$$ is Y than that of the O $$-$$ F bond in F$$_2$$O$$_2$$. Choose the correct option for X and Y from the given below.
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 36
The starting material for convenient preparation of deuterated hydrogen peroxide (D$$_2$$O$$_2$$) in laboratory is:
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 37
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A): Gypsum is used for making fireproof wall boards.
Reason (R): Gypsum is unstable at high temperatures.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 38
The correct order of bond enthalpy (kJ mol$$^{-1}$$) is:
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 39
Given below are two statements:
Statement I: Sulphanilic acid gives esterification test for carboxyl group.
Statement II: Sulphanilic acid gives red colour in Lassigne's test for extra element detection.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 40
Testosterone, which is a steroidal hormone, has the following structure.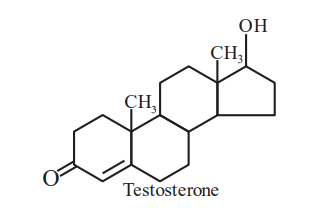
The total number of asymmetric carbon atom/s in testosterone is ______.
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 41
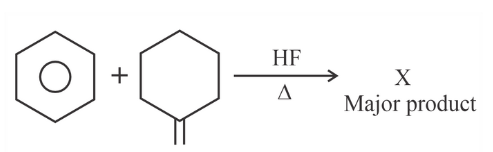
'X' is:
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 42
The industrial activity held least responsible for global warming is:
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 43
A metal M crystallizes into two lattices: face centred cubic (fcc) and body centred cubic (bcc) with unit cell edge length of $$2.0$$ and $$2.5$$ $$\text{\AA}$$ respectively. The ratio of densities of lattices fcc to bcc for the metal M is ______ (Nearest integer)
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 44
The graph which represents the following reaction is:
$$(\text{C}_6\text{H}_5)_3\text{C} - \text{Cl} \xrightarrow[\text{Pyridine}]{\text{OH}^-} (\text{C}_6\text{H}_5)_3\text{C} - \text{OH}$$
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 45
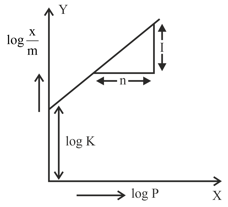
In figure, a straight line is given for Freundrich Adsorption ($$y = 3x + 2.505$$). The value of $$\frac{1}{n}$$ and log K are respectively.
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 46
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A): Cu$$^{2+}$$ in water is more stable than Cu$$^{+}$$.
Reason (R): Enthalpy of hydration for Cu$$^{2+}$$ is much less than that of Cu$$^{+}$$.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 47
Which element is not present in Nessler's reagent?
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 48
The complex cation which has two isomers is:
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 49
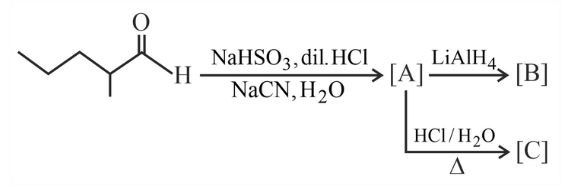
The structures of major products A, B and C in the following reaction sequence are:
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 50
In a reaction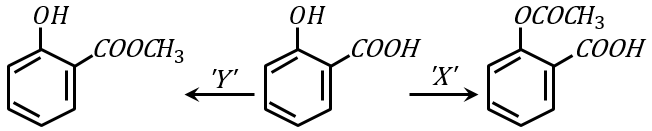
reagents 'X' and 'Y' respectively are:
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 51
All structures given below are of vitamin C. Most stable of them is:
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 52
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A): $$\alpha$$-halocarboxylic acid on reaction with dil. NH$$_3$$ gives good yield of $$\alpha$$-amino carboxylic acid whereas the yield of amines is very low when prepared from alkyl halides.
Reason (R): Amino acids exist in zwitter ion form in aqueous medium.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 53
The molality of a $$10\%(v/V)$$ solution of di-bromine solution in CCl$$_4$$ (carbon tetrachloride) is x. $$x = $$ ______ $$\times 10^{-2}$$ M. (Nearest integer)
[Given: molar mass of Br$$_2 = 160$$ g mol$$^{-1}$$, atomic mass of C $$= 12$$ g mol$$^{-1}$$, atomic mass of Cl $$= 35.5$$ g mol$$^{-1}$$, density of dibromine $$= 3.2$$ g cm$$^{-3}$$, density of CCl$$_4 = 1.6$$ g cm$$^{-3}$$]
789
456
123
0.-
Clear All
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 54
$$0.3$$ g of ethane undergoes combustion at $$27°$$C in a bomb calorimeter. The temperature of calorimeter system (including the water) is found to rise by $$0.5°$$C. The heat evolved during combustion of ethane at constant pressure is ______ kJ mol$$^{-1}$$. (Nearest integer)
[Given: The heat capacity of the calorimeter system is $$20$$ kJ K$$^{-1}$$, R $$= 8.3$$ JK$$^{-1}$$ mol$$^{-1}$$. Assume ideal gas behaviour. Atomic mass of C and H are 12 and 1 g mol$$^{-1}$$ respectively]
789
456
123
0.-
Clear All
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 55
$$20\%$$ of acetic acid is dissociated when its 5 g is added to 500 mL of water. The depression in freezing point of such water is ______ $$\times 10^{-3}$$ °C. Atomic mass of C, H and O are 12, 1 and 16 a.m.u. respectively.
[Given: Molal depression constant and density of water are $$1.86$$ K kg mol$$^{-1}$$ and $$1$$ g cm$$^{-3}$$ respectively]
789
456
123
0.-
Clear All
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 56
$$1 \times 10^{-5}$$ M AgNO$$_3$$ is added to $$1$$ L of saturated solution of AgBr. The conductivity of this solution at 298 K is _______ $$\times 10^{-8}$$ S m$$^{-1}$$.
[Given: K$$_{sp}$$(AgBr) $$= 4.9 \times 10^{-13}$$ at 298 K, $$\lambda^{0}_{\text{Ag}^+} = 6 \times 10^{-3}$$ Sm$$^2$$ mol$$^{-1}$$, $$\lambda^{0}_{\text{Br}^-} = 8 \times 10^{-3}$$ Sm$$^2$$ mol$$^{-1}$$, $$\lambda^{0}_{\text{NO}_3^-} = 7 \times 10^{-3}$$ Sm$$^2$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 57
$$\text{A} \to \text{B}$$
The above reaction is of zero order. Half life of this reaction is $$50$$ min. The time taken for the concentration of A to reduce to one-fourth of its initial value is ______ min. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 58
Among following compounds, the number of those present in copper matte is ______
A. CuCO$$_3$$
B. Cu$$_2$$S
C. Cu$$_2$$O
D. FeO
789
456
123
0.-
Clear All
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 59
The spin only magnetic moment of $$[\text{Mn}(\text{H}_2\text{O})_6]^{2+}$$ complexes is ______ B.M. (Nearest integer). (Given atomic number of Mn $$= 25$$)
789
456
123
0.-
Clear All
NTA JEE Main 1st February 2023 Shift 2 - Chemistry - Question 60
Among the following, the number of tranquilizer/s is/are ______
A. Chlorodiazepoxide
B. Veronal
C. Valium
D. Salvarsan
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




