NTA JEE Main 1st February 2023 Shift 1
For the following questions answer them individually
NTA JEE Main 1st February 2023 Shift 1 - Question 31
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: Hydrogen is an environment friendly fuel.
Reason R: Atomic number of hydrogen is 1 and it is a very light element.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 1st February 2023 Shift 1 - Question 32
Match List I with List II
(A) Slaked lime (I) NaOH
(B) Dead burnt plaster (II) Ca(OH)$$_2$$
(C) Caustic soda (III) Na$$_2$$CO$$_3$$ $$\cdot$$ 10H$$_2$$O
(D) Washing soda (IV) CaSO$$_4$$
NTA JEE Main 1st February 2023 Shift 1 - Question 33
Choose the correct statement(s):
A. Beryllium oxide is purely acidic in nature.
B. Beryllium carbonate is kept in the atmosphere of CO$$_2$$.
C. Beryllium sulphate is readily soluble in water.
D. Beryllium shows anomalous behavior.
NTA JEE Main 1st February 2023 Shift 1 - Question 34
Resonance in carbonate ion CO$$_3^{2-}$$ is
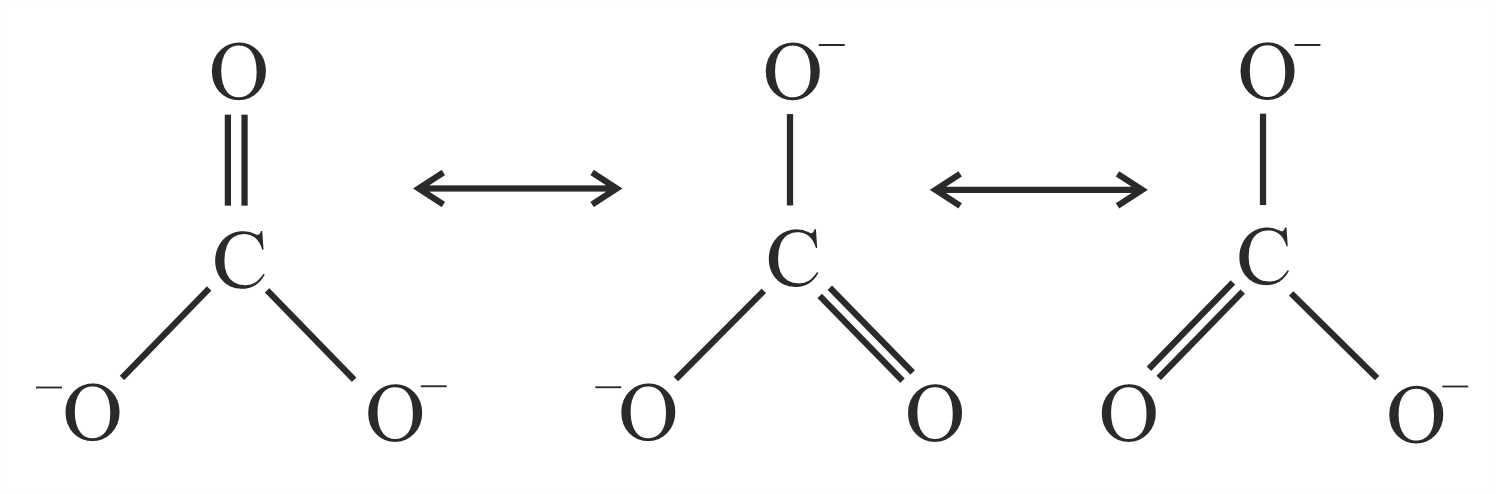
Which of the following is true?
NTA JEE Main 1st February 2023 Shift 1 - Question 35
But-2-yne is reacted separately with one mole of Hydrogen as shown below:
B $$\xleftarrow{\text{Na/liq NH}_3}$$ CH$$_3$$-C$$\equiv$$C-CH$$_3$$ $$\xrightarrow[\Delta]{\text{Pd/C, +H}_2}$$ A
Identify the incorrect statements from the options given below:
A. A is more soluble than B.
B. The boiling point & melting point of A are higher and lower than B respectively.
C. A is more polar than B because dipole moment of A is zero.
D. Br$$_2$$ adds easily to B than A.
NTA JEE Main 1st February 2023 Shift 1 - Question 36
How can photochemical smog be controlled?
NTA JEE Main 1st February 2023 Shift 1 - Question 37
Which of the following represents the lattice structure of A$$_{0.95}$$O containing A$$^{2+}$$, A$$^{3+}$$ and O$$^{2-}$$ ions?
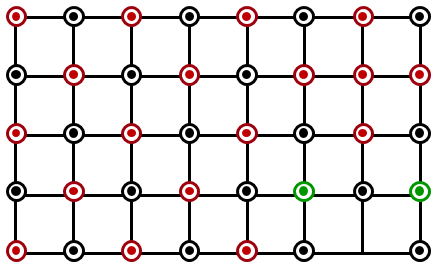
NTA JEE Main 1st February 2023 Shift 1 - Question 38
Given below are two statements:
Assertion A: Amongst He, Ne, Ar and Kr; 1 g of activated charcoal adsorbs more of Kr.
Reason R: The critical volume V$$_c$$ (cm$$^3$$ mol$$^{-1}$$) and critical pressure P$$_c$$ (atm) is highest for Krypton but the compressibility factor at critical point Z$$_c$$ is lowest for Krypton.
NTA JEE Main 1st February 2023 Shift 1 - Question 39
Given below are two statements:
Assertion A: In an Ellingham diagram, the oxidation of carbon to carbon monoxide shows a negative slope with respect to temperature.
Reason R: CO tends to get decomposed at higher temperature.
NTA JEE Main 1st February 2023 Shift 1 - Question 40
Given below are two statements:
Statement I: Chlorine can easily combine with oxygen to from oxides; and the product has a tendency to explode.
Statement II: Chemical reactivity of an element can be determined by its reaction with oxygen and halogens.


