NTA JEE Main 1st February 2023 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 31
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: Hydrogen is an environment friendly fuel.
Reason R: Atomic number of hydrogen is 1 and it is a very light element.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 32
Match List I with List II
(A) Slaked lime (I) NaOH
(B) Dead burnt plaster (II) Ca(OH)$$_2$$
(C) Caustic soda (III) Na$$_2$$CO$$_3$$ $$\cdot$$ 10H$$_2$$O
(D) Washing soda (IV) CaSO$$_4$$
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 33
Choose the correct statement(s):
A. Beryllium oxide is purely acidic in nature.
B. Beryllium carbonate is kept in the atmosphere of CO$$_2$$.
C. Beryllium sulphate is readily soluble in water.
D. Beryllium shows anomalous behavior.
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 34
Resonance in carbonate ion CO$$_3^{2-}$$ is
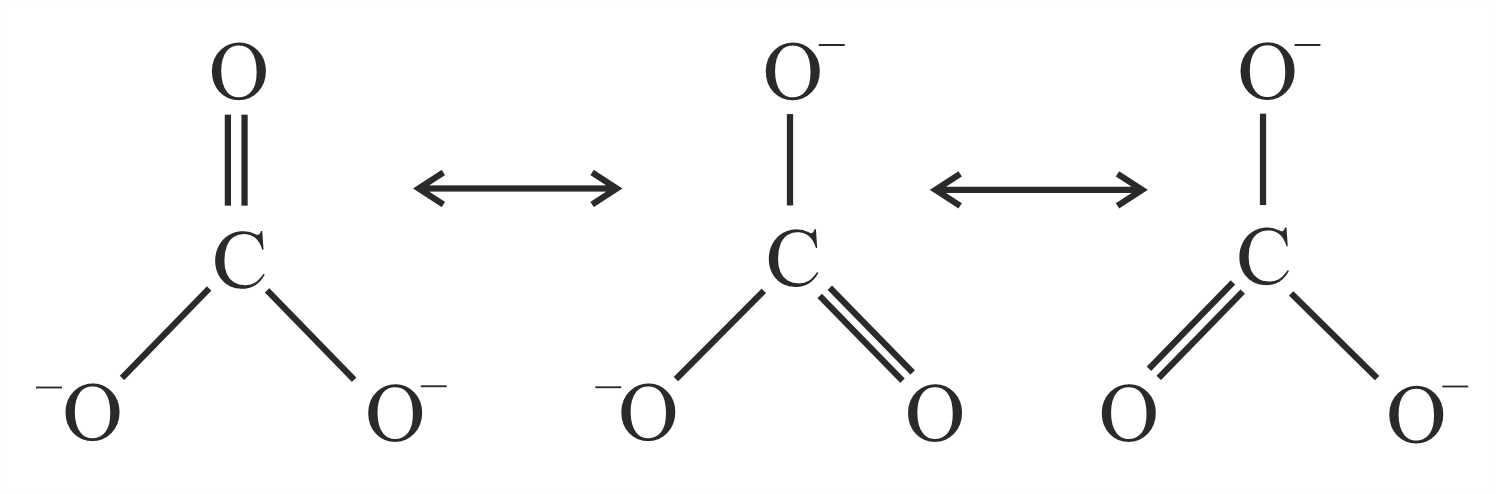
Which of the following is true?
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 35
But-2-yne is reacted separately with one mole of Hydrogen as shown below:
B $$\xleftarrow{\text{Na/liq NH}_3}$$ CH$$_3$$-C$$\equiv$$C-CH$$_3$$ $$\xrightarrow[\Delta]{\text{Pd/C, +H}_2}$$ A
Identify the incorrect statements from the options given below:
A. A is more soluble than B.
B. The boiling point & melting point of A are higher and lower than B respectively.
C. A is more polar than B because dipole moment of A is zero.
D. Br$$_2$$ adds easily to B than A.
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 36
How can photochemical smog be controlled?
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 37
Which of the following represents the lattice structure of A$$_{0.95}$$O containing A$$^{2+}$$, A$$^{3+}$$ and O$$^{2-}$$ ions?
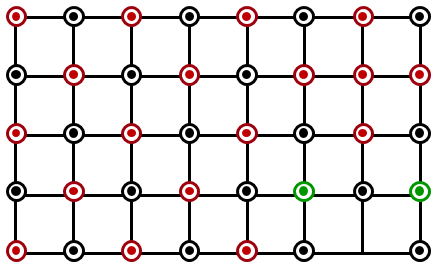
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 38
Given below are two statements:
Assertion A: Amongst He, Ne, Ar and Kr; 1 g of activated charcoal adsorbs more of Kr.
Reason R: The critical volume V$$_c$$ (cm$$^3$$ mol$$^{-1}$$) and critical pressure P$$_c$$ (atm) is highest for Krypton but the compressibility factor at critical point Z$$_c$$ is lowest for Krypton.
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 39
Given below are two statements:
Assertion A: In an Ellingham diagram, the oxidation of carbon to carbon monoxide shows a negative slope with respect to temperature.
Reason R: CO tends to get decomposed at higher temperature.
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 40
Given below are two statements:
Statement I: Chlorine can easily combine with oxygen to from oxides; and the product has a tendency to explode.
Statement II: Chemical reactivity of an element can be determined by its reaction with oxygen and halogens.
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 41
A solution of FeCl$$_3$$ when treated with K$$_4$$Fe(CN)$$_6$$ gives a prussian blue precipitate due to the formation of
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 42
Highest oxidation state of Mn is exhibited in Mn$$_2$$O$$_7$$. The correct statements about Mn$$_2$$O$$_7$$ are
(A) Mn is tetrahedrally surrounded by oxygen atoms
(B) Mn is octahedrally surrounded by oxygen atoms
(C) Contains Mn-O-Mn bridge
(D) Contains Mn-Mn bond.
Choose the correct answer from the options given below
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 43
Which of the following complex will show largest splitting of d-orbitals?
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 44
Which of the following are the example of double salt?
(A) FeSO$$_4$$ $$\cdot$$ (NH$$_4$$)$$_2$$SO$$_4$$ $$\cdot$$ 6H$$_2$$O
(B) CuSO$$_4$$ $$\cdot$$ 4NH$$_3$$ $$\cdot$$ H$$_2$$O
(C) K$$_2$$SO$$_4$$ $$\cdot$$ Al$$_2$$(SO$$_4$$)$$_3$$ $$\cdot$$ 24H$$_2$$O
(D) Fe(CN)$$_2$$ $$\cdot$$ 4KCN
Choose the correct answer.
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 45
Identify the incorrect option from the following:
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 46
Decreasing order of dehydration of the following alcohols is
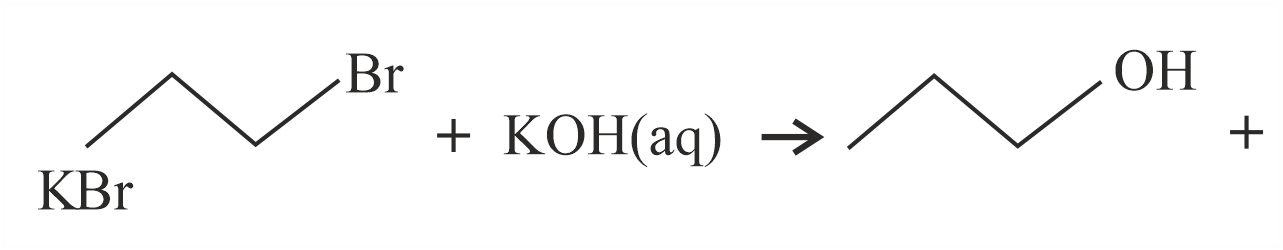
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 47
In the following reaction, 'A' is
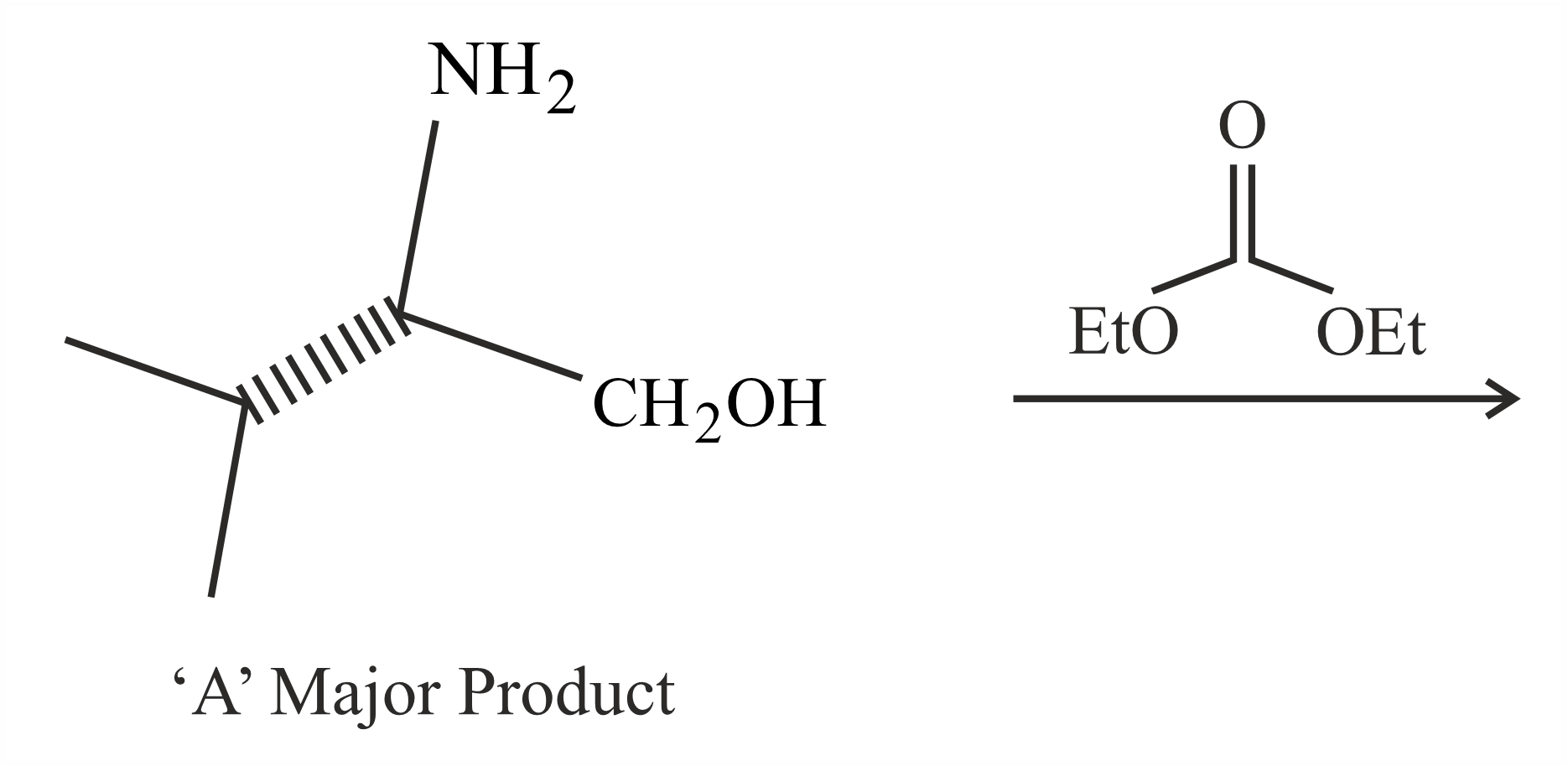
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 48
Match List I with List II
(A) Tranquilizers (I) Anti blood clotting
(B) Aspirin (II) Salvarsan
(C) Antibiotic (III) Antidepressant drugs
(D) Antiseptic (IV) Soframicine
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 49
The correct representation in six membered pyranose form for the following sugar [X] is
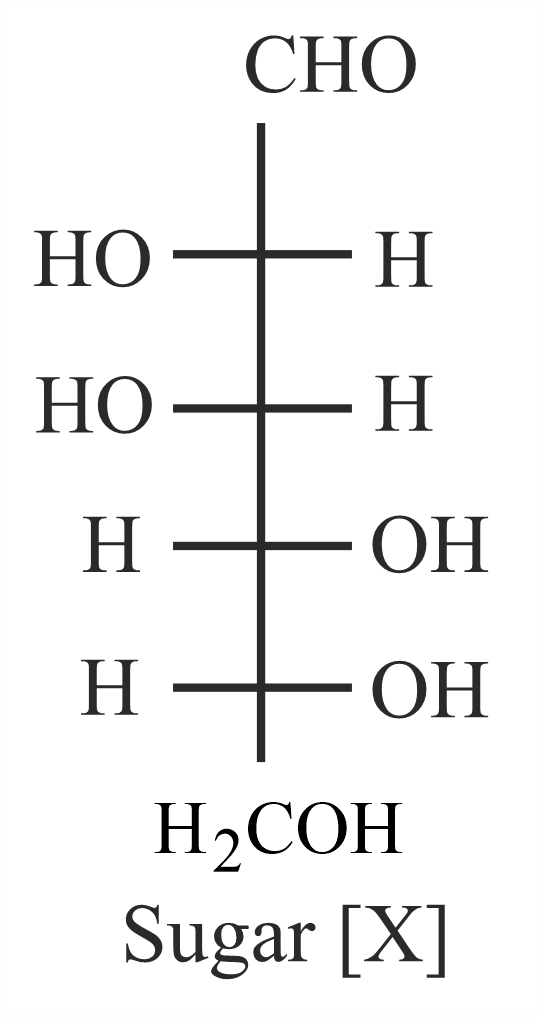
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 50
Match List I and List II
(A) Molisch's Test (I) Peptide
(B) Biuret Test (II) Carbohydrate
(C) Carbylamine Test (III) Primary amine
(D) Schiff's Test (IV) Aldehyde
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 51
The density of 3M solution of NaCl is 1.0 g mL$$^{-1}$$. Molality of the solution is _____ $$\times 10^{-2}$$ m (Nearest integer).
Given: Molar mass of Na is 23 and Cl is 35.5 g mol$$^{-1}$$ respectively.
789
456
123
0.-
Clear All
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 52
Electrons in a cathode ray tube have been emitted with a velocity of 1000 ms$$^{-1}$$. The number of following statements which is/are true about the emitted radiation is
Given: h = 6 $$\times$$ 10$$^{-34}$$Js, m$$_e$$ = 9 $$\times$$ 10$$^{-31}$$ kg
(A) The deBroglie wavelength of the electron emitted is 666.67 nm
(B) The characteristic of electrons emitted depend upon the material of the electrodes of the cathode ray tube.
(C) The cathode rays start from cathode and move towards anode.
(D) The nature of the emitted electrons depends on the nature of the gas present in cathode ray tube..
789
456
123
0.-
Clear All
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 53
At 25°C, the enthalpy of the following processes are given:
H$$_2$$(g) + O$$_2$$(g) $$\to$$ 2OH(g) $$\Delta$$H$$^0$$ = 78 kJ mol$$^{-1}$$
H$$_2$$(g) + 1/2O$$_2$$(g) $$\to$$ H$$_2$$O(g) $$\Delta$$H$$^0$$ = -242 kJ mol$$^{-1}$$
H$$_2$$(g) $$\to$$ 2H(g) $$\Delta$$H$$^0$$ = 436 kJ mol$$^{-1}$$
1/2O$$_2$$(g) $$\to$$ O(g) $$\Delta$$H$$^0$$ = 249 kJ mol$$^{-1}$$
H$$_2$$O(g) $$\to$$ Hg + OHg $$\Delta$$H$$^0$$ = X kJmol$$^{-1}$$
What would be the value of X for the following reaction? (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 54
(i) X(g) $$\rightleftharpoons$$ Y(g) + Z(g) K$$_{p1}$$ = 3
(ii) A(g) $$\rightleftharpoons$$ 2B(g) K$$_{p2}$$ = 1
If the degree of dissociation and initial concentration of both the reactants X(g) and A(g) are equal, then the ratio of the total pressure at equilibrium $$\frac{p_1}{p_2}$$ is equal to x : 1. The value of x is (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 55
The total number of chiral compound/s from the following is
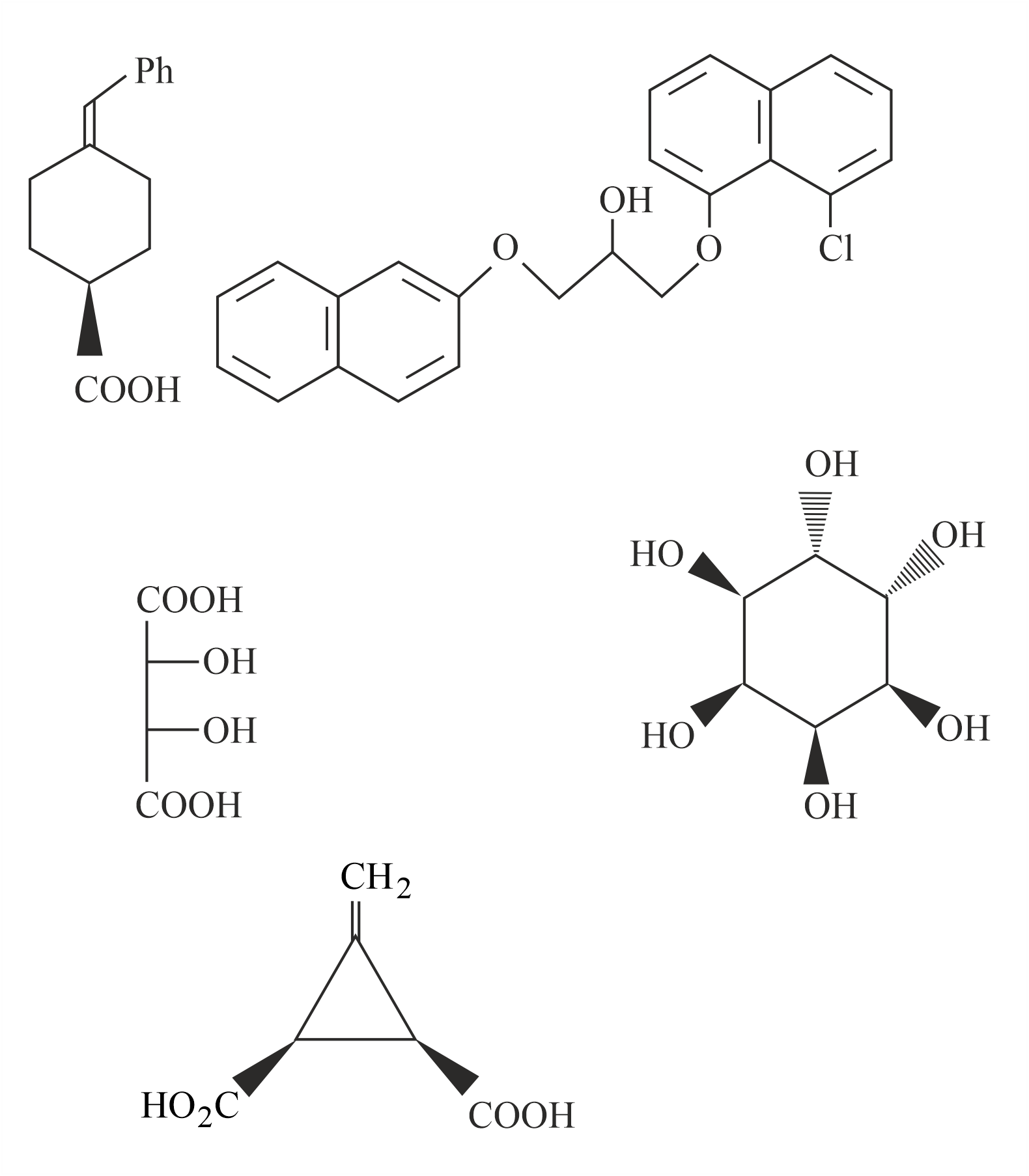
789
456
123
0.-
Clear All
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 56
25 mL of an aqueous solution of KCl was found to require 20 mL of 1M AgNO$$_3$$ solution when titrated using K$$_2$$CrO$$_4$$ as an indicator. What is the depression in freezing point of KCl solution of the given concentration?
(Nearest integer).
(Given: K$$_f$$ = 2.0 K kg mol$$^{-1}$$)
Assume 1) 100% ionization and 2) density of the aqueous solution as 1 g mL$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 57
At what pH, given half cell MnO$$_4^-$$(0.1M) | Mn$$^{2+}$$(0.001 M) will have electrode potential of 1.282 V? (Nearest Integer)
Given E$$^0_{MnO_4^-/Mn^{2+}}$$ = 1.54 V, $$\frac{2.303RT}{F}$$ = 0.059 V
789
456
123
0.-
Clear All
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 58
A and B are two substances undergoing radioactive decay in a container. The half life of A is 15 min and that of B is 5 min. If the initial concentration of B is 4 times that of A and they both start decaying at the same time, how much time will it take for the concentration of both of them to be same? _____ min.
789
456
123
0.-
Clear All
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 59
Sum of oxidation states of bromine in bromic acid and perbromic acid is
789
456
123
0.-
Clear All
NTA JEE Main 1st February 2023 Shift 1 - Chemistry - Question 60
Number of isomeric compounds with molecular formula C$$_9$$H$$_{10}$$O which
(i) do not dissolve in NaOH
(ii) do not dissolve in HCl.
(iii) do not give orange precipitate with 2, 4-DNP
(iv) on hydrogenation give identical compound with molecular formula C$$_9$$H$$_{12}$$O is
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




