JEE Advanced 2022 Paper-2
For the following questions answer them individually
JEE Advanced 2022 Paper-2 - Question 31
A double slit setup is shown in the figure. One of the slits is in medium 2 of refractive index $$n_2$$. The other slit is at the interface of this medium with another medium 1 of refractive index $$n_1$$ ($$\neq n_2$$). The line joining the slits is perpendicular to the interface and the distance between the slits is $$d$$. The slit widths are much smaller than $$d$$. A monochromatic parallel beam of light is incident on the slits from medium 1. A detector is placed in medium 2 at a large distance from the slits, and at an angle $$\theta$$ from the line joining them, so that $$\theta$$ equals the angle of refraction of the beam. Consider two approximately parallel rays from the slits received by the detector.
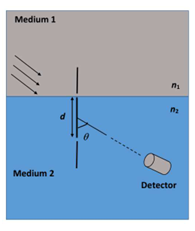
Which of the following statement(s) is (are) correct?
JEE Advanced 2022 Paper-2 - Question 32
In the given P-V diagram, a monoatomic gas $$\left(\gamma = \dfrac{5}{3}\right)$$ is first compressed adiabatically from state A to state B. Then it expands isothermally from state B to state C. [Given: $$\left(\dfrac{1}{3}\right)^{0.6} = 0.5$$, ln 2 $$\simeq$$ 0.7].
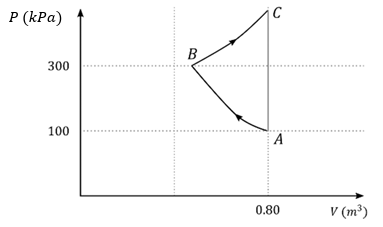
Which of the following statement(s) is(are) correct?
JEE Advanced 2022 Paper-2 - Question 33
A flat surface of a thin uniform disk A of radius R is glued to a horizontal table. Another thin uniform disk B of mass M and with the same radius R rolls without slipping on the circumference of A, as shown in the figure. A flat surface of B also lies on the plane of the table. The center of mass of B has fixed angular speed $$\omega$$ about the vertical axis passing through the center of A. The angular momentum of B is $$nM\omega R^2$$ with respect to the center of A. Which of the following is the value of $$n$$?
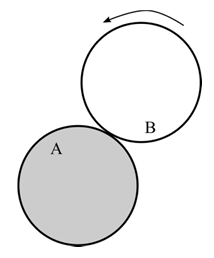
JEE Advanced 2022 Paper-2 - Question 34
When light of a given wavelength is incident on a metallic surface, the minimum potential needed to stop the emitted photoelectrons is 6.0 V. This potential drops to 0.6 V if another source with wavelength four times that of the first one and intensity half of the first one is used. What are the wavelength of the first source and the work function of the metal, respectively?
[Take $$\dfrac{hc}{e} = 1.24 \times 10^{-6}$$ J m C$$^{-1}$$.]
JEE Advanced 2022 Paper-2 - Question 35
Area of the cross-section of a wire is measured using a screw gauge. The pitch of the main scale is 0.5 mm. The circular scale has 100 divisions and for one full rotation of the circular scale, the main scale shifts by two divisions. The measured readings are listed below.
| Measurement condition | Main scale reading | Circular scale reading |
|---|---|---|
| Two arms of gauge touching each other without wire | 0 division | 4 division |
| Attempt-1: With wire | 4 divisions | 20 divisions |
| Attempt-2: With wire | 4 divisions | 16 divisions |
What are the diameter and cross-sectional area of the wire measured using the screw gauge?
JEE Advanced 2022 Paper-2 - Question 36
Which one of the following options represents the magnetic field $$\vec{B}$$ at O due to the current flowing in the given wire segments lying on the $$xy$$ plane?
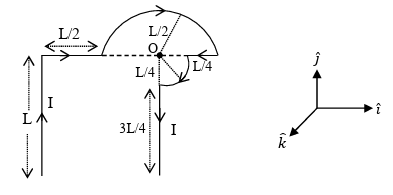
JEE Advanced 2022 Paper-2 - Question 37
Concentration of H$$_2$$SO$$_4$$ and Na$$_2$$SO$$_4$$ in a solution is 1 M and 1.8 $$\times$$ 10$$^{-2}$$ M, respectively. Molar solubility of PbSO$$_4$$ in the same solution is X $$\times$$ 10$$^{-Y}$$ M (expressed in scientific notation). The value of Y is _______.
[Given: Solubility product of PbSO$$_4$$ ($$K_{sp}$$) = 1.6 $$\times$$ 10$$^{-8}$$. For H$$_2$$SO$$_4$$, $$K_{a1}$$ is very large and $$K_{a2}$$ = 1.2 $$\times$$ 10$$^{-2}$$]
789
456
123
0.-
Clear All
JEE Advanced 2022 Paper-2 - Question 38
An aqueous solution is prepared by dissolving 0.1 mol of an ionic salt in 1.8 kg of water at 35 $$^\circ$$C. The salt remains 90% dissociated in the solution. The vapour pressure of the solution is 59.724 mm of Hg. Vapor pressure of water at 35 $$^\circ$$C is 60.000 mm of Hg. The number of ions present per formula unit of the ionic salt is _______.
789
456
123
0.-
Clear All
JEE Advanced 2022 Paper-2 - Question 39
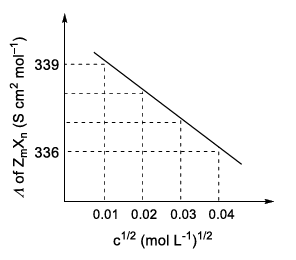
Consider the strong electrolytes Z$$_m$$X$$_n$$, U$$_m$$Y$$_p$$ and V$$_m$$X$$_n$$. Limiting molar conductivity ($$\Lambda^0$$) of U$$_m$$Y$$_p$$ and V$$_m$$X$$_n$$ are 250 and 440 S cm$$^2$$ mol$$^{-1}$$, respectively. The value of (m + n + p) is _______.
Given:
| Ion | Z$$^{n+}$$ | U$$^{p+}$$ | V$$^{n+}$$ | X$$^{m-}$$ | Y$$^{m-}$$ |
|---|---|---|---|---|---|
| $$\lambda^0$$ (S cm$$^2$$ mol$$^{-1}$$) | 50.0 | 25.0 | 100.0 | 80.0 | 100.0 |
$$\lambda^0$$ is the limiting molar conductivity of ions
The plot of molar conductivity ($$\Lambda$$) of Z$$_m$$X$$_n$$ vs c$$^{1/2}$$ is given below.
789
456
123
0.-
Clear All
JEE Advanced 2022 Paper-2 - Question 40
The reaction of Xe and O$$_2$$F$$_2$$ gives a Xe compound P. The number of moles of HF produced by the complete hydrolysis of 1 mol of P is _______.
789
456
123
0.-
Clear All



